Jorge Stolfi (talk | contribs) Merged contents of polyunsaturated fat |
Jorge Stolfi (talk | contribs) Moved back regulation to future trans fat regulation Tag: content sourced to vanity press |
||
| Line 448: | Line 448: | ||
| [[Whole Grain]] [[Wheat]] || 9.7 |
| [[Whole Grain]] [[Wheat]] || 9.7 |
||
|} |
|} |
||
==Trans fat== |
|||
[[File:Margaryn 022.jpg|thumb|upright=1.3|[[Margarine]], a common product that can contain trans fatty acids]] |
|||
{{Fats}} |
|||
'''Trans fat''', also called '''trans-unsaturated fatty acids''' or '''trans fatty acids''', is a type of [[unsaturated fat]] that occurs in small amounts in meat and milk fat.<ref>{{cite journal | vauthors = Kuhnt K, Baehr M, Rohrer C, Jahreis G | title = Trans fatty acid isomers and the trans-9/trans-11 index in fat containing foods | journal = European Journal of Lipid Science and Technology | volume = 113 | issue = 10 | pages = 1281–1292 | date = October 2011 | pmid = 22164125 | pmc = 3229980 | doi = 10.1002/ejlt.201100037 }}</ref> It became widely produced as an unintentional byproduct in the industrial processing of vegetable and fish oils in the early 20th century for use in [[margarine]]<ref>{{cite encyclopedia |last1=Gormley |first1=James J. |last2=Juturu |first2=Vijaya | name-list-format = vanc |chapter=Partially Hydrogenated Fats in the US Diet and Their Role in Disease|encyclopedia=Modern Dietary Fat Intakes in Disease Promotion |date=2010 |publisher=Humana Press | veditors = De Meester F, Zibadi S, Watson RR |location=Totowa, NJ|isbn=9781603275712 |series=Nutrition and Health |pages=85–94 |language=en |doi=10.1007/978-1-60327-571-2_5 }}</ref> and later also in snack food, packaged baked goods, and for frying fast food.<ref name=CSPI1 /><ref name="GRAS?">{{cite web|url=https://www.federalregister.gov/articles/2013/11/08/2013-26854/tentative-determination-regarding-partially-hydrogenated-oils-request-for-comments-and-for |title=Tentative Determination Regarding Partially Hydrogenated Oils |publisher=[[Federal Register]] |date=8 November 2013 |access-date=8 November 2013 |id=2013-26854, Vol. 78, No. 217 |archive-url=https://web.archive.org/web/20140406185048/https://www.federalregister.gov/articles/2013/11/08/2013-26854/tentative-determination-regarding-partially-hydrogenated-oils-request-for-comments-and-for |archive-date=6 April 2014 |url-status=live }}</ref> |
|||
Fats contain long [[hydrocarbon]] chains, which can be either unsaturated, i.e., have [[double bond]]s, or saturated, i.e., have no double bonds. In nature, unsaturated fatty acids generally have [[Cis–trans isomerism|''cis'' as opposed to ''trans'']] configurations.<ref>{{cite journal | vauthors = Martin CA, Milinsk MC, Visentainer JV, Matsushita M, de-Souza NE | title = Trans fatty acid-forming processes in foods: a review | journal = Anais da Academia Brasileira de Ciencias | volume = 79 | issue = 2 | pages = 343–50 | date = June 2007 | pmid = 17625687 | doi = 10.1590/S0001-37652007000200015 | doi-access = free }}</ref> In food production, liquid cis-unsaturated fats such as vegetable oils are [[Hydrogenation|hydrogenated]] to produce saturated fats, which have more desirable physical properties: e.g., they melt at a desirable temperature (30–40 °C; 86–104 °F); and extend the shelf-life of food. Partial hydrogenation of the unsaturated fat converts some of the cis double bonds into trans double bonds by an [[isomerization reaction]] with the [[catalyst]] used for the hydrogenation, which yields a trans fat.<ref name=CSPI1>{{cite web |url=http://cspinet.org/new/pdf/trans_q_a.pdf |title=About Trans Fat and Partially Hydrogenated Oils |publisher=[[Center for Science in the Public Interest]]}}</ref><ref name="GRAS?" /> |
|||
Although trans fats are edible, consuming trans fats has been shown to increase the risk of [[coronary artery disease]] in part by raising levels of [[low-density lipoprotein]] (LDL, often termed "bad cholesterol"), lowering levels of [[high-density lipoprotein]] (HDL, often termed "good cholesterol"), increasing triglycerides in the bloodstream and promoting systemic inflammation.<ref name=p423>{{cite book|last=Food and nutrition board, institute of medicine of the national academies|title=Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients)|publisher=National Academies Press|year=2005|pages=[https://archive.org/details/isbn_9780309085250/page/423 423]|url=https://archive.org/details/isbn_9780309085250/page/423|doi=10.17226/10490|isbn=978-0-309-08525-0}}</ref><ref>{{cite web|title=Trans fat: Avoid this cholesterol double whammy|publisher=Mayo Foundation for Medical Education and Research (MFMER)|url=http://www.mayoclinic.com/health/trans-fat/CL00032|access-date=10 December 2007}}</ref> |
|||
Trans fats also occur naturally, e.g., the [[vaccenic acid]] in breast milk, and some [[isomers]] of [[conjugated linoleic acid]] (CLA). These trans fats occur naturally in meat and dairy products from [[ruminant]]s. Butter, for example, contains about 3% trans fat.<ref>{{cite web |url=https://ndb.nal.usda.gov/ndb/foods/show/1?fgcd=&manu=&lfacet=&format=&count=&max=50&offset=50&sort=default&order=asc&qlookup=butter&ds=&qt=&qp=&qa=&qn=&q=&ing= |title=National Nutrient Database for Standard Reference Release 28 |publisher=[[United States Department of Agriculture]]}}{{Dead link|date=August 2018 |bot=InternetArchiveBot |fix-attempted=yes}}</ref> Two Canadian studies have shown that vaccenic acid could be beneficial compared to hydrogenated vegetable [[shortening]], or a mixture of pork lard and soy fat, by lowering total LDL and triglyceride levels.<ref>[http://www.redorbit.com/news/health/2608879/trans-fats-from-ruminant-animals-may-be-beneficial Trans Fats From Ruminant Animals May Be Beneficial – Health News]. redOrbit (8 September 2011). Retrieved 22 January 2013.</ref><ref name="jn.nutrition">{{cite journal | vauthors = Bassett CM, Edel AL, Patenaude AF, McCullough RS, Blackwood DP, Chouinard PY, Paquin P, Lamarche B, Pierce GN | display-authors = 6 | title = Dietary vaccenic acid has antiatherogenic effects in LDLr-/- mice | journal = The Journal of Nutrition | volume = 140 | issue = 1 | pages = 18–24 | date = January 2010 | pmid = 19923390 | doi = 10.3945/jn.109.105163 | doi-access = free }}</ref><ref>{{cite journal|vauthors=Wang Y, Jacome-Sosa MM, Vine DF, Proctor SD |title=Beneficial effects of vaccenic acid on postprandial lipid metabolism and dyslipidemia: Impact of natural trans-fats to improve CVD risk|date=20 May 2010|journal=Lipid Technology|volume=22|issue=5|pages=103–106|doi=10.1002/lite.201000016}}</ref><ref name="jn.nutrition"/> A study by the US Department of Agriculture showed that vaccenic acid raises both HDL and LDL cholesterol, whereas industrial trans fats only raise LDL with no beneficial effect on HDL.<ref>David J. Baer, PhD. US Department of Agriculture, Agricultural Research Service, Beltsville Human Nutrition Research Laboratory. [https://web.archive.org/web/20130208183218/http://wds2010.com/delegates/presentations/10wed/06-Session0_2-David%20Baer.pdf ''New Findings on Dairy Trans Fat and Heart Disease Risk''], IDF World Dairy Summit 2010, 8–11 November 2010. Auckland, New Zealand</ref> |
|||
In light of recognized evidence and scientific agreement, nutritional authorities consider all trans fats equally harmful for health and recommend that their consumption be reduced to trace amounts.<ref>{{cite journal|last=EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA)|title=Scientific opinion on dietary reference values for fats|doi=10.2903/j.efsa.2010.1461|journal=EFSA Journal|year=2010|volume=8|issue=3|pages = 1461|doi-access=free}}</ref><ref>{{cite web|title=Update on trans fatty acids and health, Position Statement|last=UK Scientific Advisory Committee on Nutrition|year=2007|archive-url=http://webarchive.nationalarchives.gov.uk/20101210102056/http://www.food.gov.uk/multimedia/pdfs/board/fsa071207.pdf|url=http://www.food.gov.uk/multimedia/pdfs/board/fsa071207.pdf|archive-date=10 December 2010|url-status=dead}}</ref><ref>{{cite journal | vauthors = Brouwer IA, Wanders AJ, Katan MB | title = Effect of animal and industrial trans fatty acids on HDL and LDL cholesterol levels in humans--a quantitative review | journal = PLOS ONE | volume = 5 | issue = 3 | pages = e9434 | date = March 2010 | pmid = 20209147 | pmc = 2830458 | doi = 10.1371/journal.pone.0009434 | bibcode = 2010PLoSO...5.9434B }}</ref><ref>{{cite web|url=http://www.hc-sc.gc.ca/hl-vs/iyh-vsv/food-aliment/trans-eng.php |website=It's your health |publisher=Health Canada |title=Trans fat |date=Dec 2007 |url-status=dead |archive-url=https://web.archive.org/web/20120420052235/http://www.hc-sc.gc.ca/hl-vs/iyh-vsv/food-aliment/trans-eng.php |archive-date=20 April 2012}}</ref><ref>{{cite press release|title=EFSA sets European dietary reference values for nutrient intakes|url=http://www.efsa.europa.eu/en/press/news/nda100326 |publisher=European Food Safety Authority|date=26 March 2010}}</ref> The [[World Health Organization]] recommended that trans fats make up no more than 0.9% of a person's diet in 2003<ref name=Canada/> and, in 2018, introduced a 6-step guide to eliminate industrially-produced trans-fatty acids from the global food supply.<ref>{{cite press release|title=WHO plan to eliminate industrially-produced trans-fatty acids from global food supply|url=http://www.who.int/news-room/detail/14-05-2018-who-plan-to-eliminate-industrially-produced-trans-fatty-acids-from-global-food-supply |publisher=World Health Organization|date=14 May 2018}}</ref> |
|||
In many countries, there are legal limits to trans fat content. Trans fats levels can be reduced or eliminated by switching to saturated fats such as [[lard]], [[palm oil]], or fully hydrogenated fats, or by using [[interesterified fat]]. Other alternative formulations can also allow unsaturated fats to be used to replace saturated or partially hydrogenated fats. Hydrogenated oil is not a synonym for trans fat: complete hydrogenation removes all unsaturated fats. |
|||
{{TOC limit|3}} |
|||
===History=== |
|||
[[File:Crisco Cookbook 1912.jpg|thumb|Cover of original Crisco cookbook, 1912]] |
|||
[[Nobel prize|Nobel]] [[laureate]] [[Paul Sabatier (chemist)|Paul Sabatier]] worked in the late 1890s to develop the chemistry of hydrogenation, which enabled the [[margarine]], oil hydrogenation, and synthetic [[methanol]] industries.<ref>{{cite book|title=Nobel Lectures, Chemistry, 1901–1921|year=1966|publisher=Elsevier}} Reprinted online: {{cite web|title=Paul Sabatier, The Nobel Prize in Chemistry 1912|publisher=Nobel Foundation|url=http://nobelprize.org/nobel_prizes/chemistry/laureates/1912/sabatier-bio.html|access-date=7 January 2007}}</ref> Whereas Sabatier considered hydrogenation of only vapors, the German [[chemist]] [[Wilhelm Normann]] showed in 1901 that liquid oils could be hydrogenated, and patented the process in 1902.<ref>{{patent|de|141029|Process for converting unsaturated fatty acids or their glycerides into saturated compounds}}</ref><ref>{{patent|gb|190301515|Process for converting unsaturated fatty acids or their glycerides into saturated compounds}}</ref><ref>{{cite journal| vauthors = Patterson HB |title=Hydrogenation |journal=Sci Lecture Papers Series |year=1998 |url=http://www.soci.org/SCI/publications/2001/pdf/pb97.pdf |access-date=7 January 2007 |url-status=dead |archive-url=https://web.archive.org/web/20070926141346/http://www.soci.org/SCI/publications/2001/pdf/pb97.pdf |archive-date=26 September 2007}}</ref> During the years 1905–1910, Normann built a fat-hardening facility in the [[Herford]] company. At the same time, the invention was extended to a large-scale plant in [[Warrington]], England, at [[Joseph Crosfield|Joseph Crosfield & Sons, Limited]]. It took only two years until the hardened fat could be successfully produced in the plant in Warrington, commencing production in the autumn of 1909. The initial year's production totalled nearly 3,000 tonnes.<ref name=r1>{{cite web|url=http://www.dgfett.de/history/normann/nr_fiedler.htm|title=Wilhelm Normann und die Geschichte der Fetthärtung von Martin Fiedler, 2001|date=20 December 2011|access-date=14 August 2007|archive-url=https://web.archive.org/web/20111001212626/http://www.dgfett.de/history/normann/nr_fiedler.htm|archive-date=1 October 2011|url-status=dead|df=dmy-all}}</ref> |
|||
In 1909, [[Procter & Gamble]] acquired the United States rights to the Normann patent;<ref>{{cite web| last1=Shurtleff|first1=William|last2=Aoyagi|first2=Akiko| name-list-format = vanc |title=History of Soybeans and Soyfoods: 1100 B.C. to the 1980s|archive-url=https://web.archive.org/web/20051018105337/http://www.thesoydailyclub.com/SFC/MSPproducts501.asp|url=http://www.thesoydailyclub.com/SFC/MSPproducts501.asp|archive-date=18 October 2005}}</ref> in 1911, they began marketing the first hydrogenated [[shortening]], [[Crisco]] (composed largely of partially hydrogenated [[cottonseed oil]]). Further success came from the marketing technique of giving away free cookbooks in which every recipe called for Crisco. |
|||
Normann's hydrogenation process made it possible to stabilize affordable [[whale oil]] or [[fish oil]] for human consumption, a practice kept secret to avoid consumer distaste.<ref name=r1/> |
|||
Before 1910, dietary fats in industrialized nations consisted mostly of [[butterfat]], beef [[tallow]], and [[lard]]. During Napoleon's reign in France in the early 19th century, a type of margarine was invented to feed the troops using tallow and buttermilk. It was not accepted in the United States. In the early 20th century, soybeans began to be imported into the United States as a source of protein; soybean oil was a by-product. What to do with that oil became an issue. At the same time, there was not enough butterfat available for consumers. The method of hydrogenating fat and turning a liquid fat into a solid one had been discovered, and now the ingredients (soybeans) and the ''need'' (shortage of butter) were there. Later, the means for storage, the refrigerator, was a factor in trans fat development. The fat industry found that hydrogenated fats provided some special features to margarines, which allowed margarine, unlike butter, to be taken out of a refrigerator and immediately spread on bread. By some minor changes to the chemical composition of hydrogenated fat, such hydrogenated fat was found to provide superior baking properties compared to lard. Margarine made from hydrogenated soybean oil began to replace butterfat. Hydrogenated fat such as Crisco and [[Spry Vegetable Shortening|Spry]], sold in England, began to replace butter and lard in baking bread, pies, cookies, and cakes in 1920.<ref>{{cite book | first = Fred A. | last = Kummerow | name-list-format = vanc |title=Cholesterol Won't Kill You – But Trans Fat Could|publisher=Trafford Publishing|year=2008|isbn=978-1-4251-3808-0}}</ref> |
|||
Production of hydrogenated fats increased steadily until the 1960s, as processed vegetable fats replaced animal fats in the United States and other Western countries. At first, the argument was a financial one due to lower costs; advocates also said that the unsaturated trans fats of margarine were healthier than the [[saturated fat]]s of butter.<ref name="Harvard">{{cite web|vauthors=Ascherio A, Stampfer MJ, Willett WC |title=Trans fatty acids and coronary heart disease|url=http://www.hsph.harvard.edu/reviews/transfats.html|access-date=14 September 2006|archive-url=https://web.archive.org/web/20060903061226/http://www.hsph.harvard.edu/reviews/transfats.html|archive-date=3 September 2006}}</ref> |
|||
A [[University of Guelph]] research group has found a way to mix oils (such as olive, soybean, and canola), water, [[monoglyceride]]s, and fatty acids to form a "cooking fat" that acts the same way as trans and saturated fats.<ref>{{cite news|url=http://www.cbc.ca/health/fitness-blog/2007/01/trans_fats_headed_for_the_exit.html|title=Trans Fats Headed for the Exit| last = Hadzipetros | first = Peter | name-list-format = vanc |date=25 January 2007|publisher=CBC News}}</ref><ref>{{cite web|url=http://www.rsc.org/Publishing/ChemScience/Volume/2007/02/trans_fat_free_future.asp|title=Trans fat free future|website=Royal Society of Chemistry| last = Spencelayh | first = Michael | name-list-format = vanc |date=9 January 2007}}</ref> |
|||
===Chemistry=== |
|||
In chemical terms, ''trans fat'' is a fat (lipid) molecule that contains one or more double bonds in ''trans'' geometric configuration. |
|||
A double bond may exhibit one of two possible configurations: ''trans'' or ''cis''. In ''trans'' configuration, the carbon chain extends from opposite sides of the double bond, whereas, in ''cis'' configuration, the carbon chain extends from the same side of the double bond. The ''trans'' molecule is a straighter molecule. The ''cis'' molecule is bent.<ref>Udo Erasmus; ''Fats that heal, Fats that Kill'', Alive books, 1993 edition, Pages 13-19.</ref> |
|||
{|class="wikitable" style="text-align:center; margin:auto;" |
|||
|- |
|||
! style="width:250px;"|Trans ([[Elaidic acid]]) |
|||
! style="width:250px;"|Unsaturated ([[Oleic acid]]) |
|||
! style="width:250px;"|Saturated ([[Stearic acid]]) |
|||
|- |
|||
| Elaidic acid is the main ''trans'' unsaturated fatty acid often found in partially hydrogenated vegetable oils.<ref name=Alonso>{{cite journal | vauthors = Alonso L, Fontecha J, Lozada L, Fraga MJ, Juárez M | title = Fatty acid composition of caprine milk: major, branched-chain, and trans fatty acids | journal = Journal of Dairy Science | volume = 82 | issue = 5 | pages = 878–84 | date = May 1999 | pmid = 10342226 | doi = 10.3168/jds.S0022-0302(99)75306-3 | hdl = 10261/113439 | doi-access = free }}</ref> |
|||
| Oleic acid is an unsaturated fatty acid making up 55–80% of olive oil.<ref>{{cite encyclopedia| first = Alfred | last = Thomas | name-list-format = vanc |title=Ullmann's Encyclopedia of Industrial Chemistry|encyclopedia=Ullmann's Encyclopedia of Industrial Chemistry|publisher=Wiley-VCH|place=Weinheim|year=2002|doi=10.1002/14356007.a10_173|chapter=Fats and Fatty Oils|isbn=978-3-527-30673-2}}</ref> |
|||
| Stearic acid is a saturated fatty acid found in animal fats and is the intended product in full hydrogenation. Stearic acid is neither ''unsaturated'' nor ''trans'' because it has no carbon-carbon double bonds.<!--a spacefill version of this one would work much better --> |
|||
|- style="background:white;" |
|||
| [[File:Elaidic-acid-3D-vdW.png|center|250px]] |
|||
| [[File:Oleic-acid-3D-vdW.png|center|250px]] |
|||
| [[File:Stearic-acid-3D-balls.png|250px]] |
|||
|- style="background:white;" |
|||
| [[File:Elaidic-acid-2D-skeletal.png|center|250px]] |
|||
| [[File:Oleic-acid-skeletal.svg|center|250px]] |
|||
| [[File:Stearic acid.svg|center|250px]] |
|||
|- |
|||
| colspan=2|These fatty acids are [[geometric isomer]]s (structurally identical except for the arrangement of the double bond). |
|||
| This fatty acid contains no carbon-carbon double bonds and is ''not'' isomeric with the prior two. |
|||
|} |
|||
A fatty acid is characterized as either ''saturated'' or ''unsaturated'' based on the presence of double bonds in its structure. If the molecule contains no double bonds, it is said to be saturated; otherwise, it is unsaturated to some degree.<ref>{{cite book|title=Food lipids: chemistry, nutrition, and biotechnology|pages=1–2|isbn=978-0-8247-0749-1|editor1=Casimir C. Akoh |editor2=David B. Min.|year=2002|publisher=M. Dekker|location=New York}}</ref><ref>{{cite book|chapter-url=http://goldbook.iupac.org/F02330.html|chapter="fatty acid"|title=IUPAC Gold book|publisher=[[International Union of Pure and Applied Chemistry]]}}</ref> |
|||
Only unsaturated fats can be ''trans'' ''or'' ''cis'' fat, since only a double bond can be locked to these orientations. Saturated fatty acids are never called ''trans fats'' because they have no double bonds. Thus, all their bonds are freely rotatable. Other types of fatty acids, such as [[crepenynic acid]], which contains a [[triple bond]], are rare and of no nutritional significance.{{citation needed|date=September 2015|reason=How do we know triple bonds are rare and don't matter?}} |
|||
Carbon atoms are [[tetravalent]], forming four [[covalent bond]]s with other atoms, whereas hydrogen atoms bond with only one other atom. In saturated fatty acids, each carbon atom (besides the last) is connected to its two neighbour carbon atoms and to two hydrogen atoms. In unsaturated fatty acids, the carbon atoms that are missing a hydrogen atom are joined by double bonds rather than single bonds so that each carbon atom still participates in four bonds. |
|||
[[File:Wilhelm Normann.jpg|thumb|[[Wilhelm Normann]] patented the hydrogenation of liquid oils in 1902]] |
|||
[[Hydrogenation]] of an unsaturated fatty acid refers to the addition of hydrogen atoms to the acid, causing double bonds to become single ones, as carbon atoms acquire new hydrogen partners (to maintain four bonds per carbon atom). Full hydrogenation results in a molecule containing the maximum amount of hydrogen (in other words, the conversion of an unsaturated fatty acid into a saturated one). Partial hydrogenation results in the addition of hydrogen atoms at some of the empty positions, with a corresponding reduction in the number of double bonds. Typical commercial hydrogenation is partial to obtain a [[malleable]] mixture of fats that is solid at [[room temperature]], but melts during baking, or consumption. |
|||
In most naturally occurring unsaturated fatty acids, the hydrogen atoms are on the same side of the double bonds of the carbon chain (''[[Cis-trans isomerism|cis]]'' configuration – from the Latin, meaning "on the same side"). However, partial hydrogenation reconfigures most of the double bonds that do not become chemically saturated, twisting them so that the hydrogen atoms end up on different sides of the chain. This type of configuration is called ''[[Cis-trans isomerism|trans]]'', from the Latin, meaning "across".<ref>{{cite book|last1=Hill|first1=John W. |last2=Kolb|first2=Doris K | name-list-format = vanc |isbn=978-0136054498|title=Chemistry for changing times|publisher=Pearson / Prentice Hall|year=2007}}</ref> The trans configuration is the lower energy form, and is favored when catalytically equilibrated as a side reaction in hydrogenation. |
|||
The same molecule, containing the same number of atoms, with a double bond in the same location, can be either a ''trans'' or a ''cis'' fatty acid depending on the configuration of the double bond. For example, [[oleic acid]] and [[elaidic acid]] are both unsaturated fatty acids with the [[chemical formula]] C<sub>9</sub>H<sub>17</sub>C<sub>9</sub>H<sub>17</sub>O<sub>2</sub>.<ref name="Taylor" /> They both have a double bond located midway along the carbon chain. It is the configuration of this bond that sets them apart. The configuration has implications for the [[physical chemistry|physical-chemical]] properties of the molecule. The ''trans'' configuration is straighter, while the ''cis'' configuration is noticeably kinked as can be seen from the three-dimensional representation shown above. |
|||
The ''trans'' fatty acid elaidic acid has different chemical and physical properties, owing to the slightly different bond configuration. It has a much higher melting point, 45 [[Celsius|°C]], than oleic acid, 13.4 °C, due to the ability of the trans molecules to pack more tightly, forming a solid that is more difficult to break apart.<ref name="Taylor">{{cite book|title=Handbook of chemistry and physics|url=http://www.hbcpnetbase.com|access-date=19 November 2007|edition=88th|series=2007–2008|year=2007|publisher=Taylor and Francis|chapter=Section 7: Biochemistry|chapterurl=http://www.hbcpnetbase.com/articles/07_05_86.pdf}}</ref> This notably means that it is a solid at human body temperatures. |
|||
In the sense of food production, however, the goal is not necessarily to simply change the configuration of double bonds while maintaining the same ratios of hydrogen to carbon; rather, the goal is to decrease the number of double bonds (when a fatty acid molecule contains more than one double bond it is classified as "polyunsaturated") by increasing the amount of hydrogen (and, thus, single bonds) in the fatty acid. This subsequent lesser degree of unsaturation (and, simultaneously, greater degree of saturation) thereby changes the consistency of the fatty acid by way of allowing its molecules to more greatly compress and congeal and in turn thereby makes it less prone to [[rancidity]] (in which free [[Radical (chemistry)|radicals]] attack double bonds). In this second sense of the goal being to simply reduce the degree of unsaturation in an unsaturated fatty acid, the production of trans fatty acids is thus an undesirable side effect of partial hydrogenation. |
|||
Catalytic partial hydrogenation necessarily produces ''trans''-fats, because of the [[Hydrogenation#Heterogeneous catalysis|reaction mechanism]]. In the first reaction step, one hydrogen is added, with the other, coordinatively unsaturated, carbon being attached to the catalyst. The second step is the addition of hydrogen to the remaining carbon, producing a saturated fatty acid. The first step is reversible, such that the hydrogen is readsorbed on the catalyst and the double bond is re-formed. The intermediate with only one hydrogen added contains no double bond and can freely rotate. Thus, the double bond can re-form as either ''cis'' or ''trans'', of which ''trans'' is favored, regardless the starting material. Complete hydrogenation also hydrogenates any produced ''trans'' fats to give saturated fats. |
|||
Researchers at the [[United States Department of Agriculture]] have investigated whether hydrogenation can be achieved without the side effect of trans fat production. They varied the [[pressure]] under which the chemical reaction was conducted – applying 1400 [[Pascal (unit)|kPa]] (200 [[pound-force per square inch|psi]]) of pressure to soybean oil in a 2-liter vessel while heating it to between 140 °C and 170 °C. The standard 140 kPa (20 psi) process of hydrogenation produces a product of about 40% trans fatty acid by weight, compared to about 17% using the high-pressure method. Blended with unhydrogenated liquid soybean oil, the high-pressure-processed oil produced margarine containing 5 to 6% trans fat. Based on current U.S. labeling requirements (see below), the manufacturer could claim the product was free of trans fat.<ref>{{cite journal | vauthors = Eller FJ, List GR, Teel JA, Steidley KR, Adlof RO | title = Preparation of spread oils meeting U.S. Food and Drug Administration Labeling requirements for trans fatty acids via pressure-controlled hydrogenation | journal = Journal of Agricultural and Food Chemistry | volume = 53 | issue = 15 | pages = 5982–4 | date = July 2005 | pmid = 16028984 | doi = 10.1021/jf047849+ }}</ref> The level of trans fat may also be altered by modification of the temperature and the length of time during hydrogenation. |
|||
Trans fat levels may be measured. Measurement techniques include [[chromatography]] (by [[silver ion chromatography]] on [[thin layer chromatography]] plates, or small [[high-performance liquid chromatography]] columns of [[silica gel]] with bonded phenylsulfonic acid groups whose hydrogen atoms have been exchanged for silver ions). The role of silver lies in its ability to form complexes with unsaturated compounds. [[Gas chromatography]] and mid-[[infrared spectroscopy]] are other methods in use. |
|||
===Presence in food=== |
|||
{| class="wikitable" |
|||
|+ Trans fat contents in various foods, ranked in g per 100 g<ref>{{cite journal | vauthors = Tarrago-Trani MT, Phillips KM, Lemar LE, Holden JM | title = New and existing oils and fats used in products with reduced trans-fatty acid content | journal = Journal of the American Dietetic Association | volume = 106 | issue = 6 | pages = 867–80 | date = June 2006 | pmid = 16720128 | doi = 10.1016/j.jada.2006.03.010 }}</ref> |
|||
|- |
|||
! Food type |
|||
! Trans fat content |
|||
|- |
|||
| shortenings |
|||
| 10g to 33 g |
|||
|- |
|||
| margarine, spreads |
|||
| 0.2<ref name=BvM>{{cite news|title=Heart Foundation: Butter has 20 times the trans fats of marg {{!}} Australian Food News|url=http://www.ausfoodnews.com.au/2010/09/28/heart-foundation-butter-has-20-times-the-trans-fats-of-marg.html|work=www.ausfoodnews.com.au}}</ref> to 26 g |
|||
|- |
|||
| butter |
|||
|2g to 7 g |
|||
|- |
|||
| whole milk |
|||
|0.07g to 0.1 g |
|||
|- |
|||
| breads/cake products |
|||
| 0.1g to 10 g |
|||
|- |
|||
| cookies and crackers |
|||
| 1g to 8 g |
|||
|- |
|||
| salty snacks |
|||
| 0g to 4 g |
|||
|- |
|||
| cake frostings, sweets |
|||
| 0.1g to 7 g |
|||
|- |
|||
| animal fat |
|||
| 0g to 5 g<ref name=Canada/> |
|||
|- |
|||
|ground beef |
|||
|1 g |
|||
|} |
|||
A type of trans fat occurs naturally in the milk and body fat of [[ruminant]]s (such as cattle and sheep) at a level of 2–5% of total fat.<ref name=Canada>{{cite book|isbn=0-662-43689-X|author=Trans Fat Task Force|title=TRANSforming the Food Supply|date=June 2006|url=http://www.hc-sc.gc.ca/fn-an/nutrition/gras-trans-fats/tf-ge/tf-gt_rep-rap_e.html|access-date=7 January 2007}}</ref> Natural ''trans'' fats, which include [[conjugated linoleic acid]] (CLA) and [[vaccenic acid]], originate in the [[rumen]] of these animals. CLA has two double bonds, one in the ''cis'' configuration and one in ''trans'', which makes it simultaneously a ''cis''- and a ''trans''-fatty acid.{{<ref>[https://mejorandomishabitos.es/dieta-detox/#_DIETA_DETOX_para_Reducir_Colesterol_,_Trigliceridos,_Tension_sanguinea_y_emocional_(previniendo_la_primera_y_segunda_causas_de_muerte_en_nuestro_mundo_industrializado) trans-fatty acid]</ref>}} |
|||
Animal-based fats were once the only ''trans'' fats consumed, but by far the largest amount of ''trans'' fat consumed today is created by the processed food industry as a side effect of partially hydrogenating unsaturated plant fats (generally vegetable oils). These partially hydrogenated fats have displaced natural solid fats and liquid oils in many areas, the most notable ones being in the fast food, [[snack food]], fried food, and baked goods industries.<ref name=Ashok>{{cite book |last1=Ashok |first1=Chauhan |last2=Ajit |first2=Varma | name-list-format = vanc |date=2009 |pages = 181 |chapter=Chapter 4: Fatty acids |title=A Textbook of Molecular Biotechnology |isbn=978-93-80026-37-4}}</ref> |
|||
Partially hydrogenated oils have been used in food for many reasons. Hydrogenation increases product [[shelf life]] and decreases refrigeration requirements. Many baked foods require semi-solid fats to suspend solids at room temperature; partially hydrogenated oils have the right consistency to replace animal fats such as butter and [[lard]] at lower cost. They are also an inexpensive alternative to other semi-solid oils such as [[palm oil]].{{citation needed|date=June 2015}} |
|||
[[File:Triglyceride Hydrogenation&Isomerization V.1.png|thumb|330px|right|Reaction scheme: By far the largest amount of ''trans'' fat consumed today is created by the processed food industry as a side effect of partially catalytic hydrogenation of unsaturated plant fats (generally vegetable oils) with ''cis'' carbon-carbon double bonds. These partially hydrogenated fats have displaced natural solid fats and liquid oils in many areas, the most notable ones being in the fast food, [[snack food]], fried food, and baked goods industries.]] |
|||
Up to 45% of the total fat in those foods containing man-made ''trans'' fats formed by partially hydrogenating plant fats may be ''trans'' fat.<ref name=Canada/> Baking shortenings, unless reformulated, contain around 30% ''trans'' fats compared to their total fats. High-fat dairy products such as butter contain about 4%. Margarines not reformulated to reduce ''trans'' fats may contain up to 15% ''trans'' fat by weight,<ref name=Hunter>{{cite journal| vauthors = Hunter JE | year=2005|title=Dietary levels of trans fatty acids" basis for health concerns and industry efforts to limit use|journal=Nutrition Research|volume=25|pages=499–513|doi=10.1016/j.nutres.2005.04.002|issue=5}}</ref> but some reformulated ones are less than 1% trans fat. |
|||
It has been established that ''trans'' fats in human [[breast milk]] fluctuate with maternal consumption of trans fat, and that the amount of trans fats in the bloodstream of breastfed infants fluctuates with the amounts found in their milk. In 1999, reported percentages of trans fats (compared to total fats) in human milk ranged from 1% in Spain, 2% in France, 4% in Germany, and 7% in Canada and the United States.<ref>{{cite journal | vauthors = Innis SM, King DJ | title = trans Fatty acids in human milk are inversely associated with concentrations of essential all-cis n-6 and n-3 fatty acids and determine trans, but not n-6 and n-3, fatty acids in plasma lipids of breast-fed infants | journal = The American Journal of Clinical Nutrition | volume = 70 | issue = 3 | pages = 383–90 | date = September 1999 | pmid = 10479201 | doi = 10.1093/ajcn/70.3.383 | doi-access = free }}</ref> |
|||
Trans fats are used in shortenings for deep-frying in restaurants, as they can be used for longer than most conventional oils before becoming rancid. In the early 21st century, non-hydrogenated vegetable oils that have lifespans exceeding that of the frying shortenings became available.<ref>{{cite web|last=NYC Board of Health |title=Board of Health Approves Regulation to Phase Out Artificial Trans Fat: FAQ |url=http://www.nyc.gov/html/doh/html/cardio/cardio-transfat-healthcode-faq.shtml#7 |access-date=7 January 2007 |url-status=dead |archive-url=https://web.archive.org/web/20061006022908/http://www.nyc.gov/html/doh/html/cardio/cardio-transfat-healthcode-faq.shtml |archive-date=6 October 2006}}</ref> As fast-food chains routinely use different fats in different locations, trans fat levels in fast food can have large variations. For example, an analysis of samples of [[McDonald's]] French fries collected in 2004 and 2005 found that fries served in New York City contained twice as much trans fat as in [[Hungary]], and 28 times as much as in [[Denmark]], where trans fats are restricted. At KFC, the pattern was reversed, with Hungary's product containing twice the trans fat of the New York product. Even within the United States there was variation, with fries in New York containing 30% more trans fat than those from [[Atlanta]].<ref>{{cite news|title=What's in that french fry? Fat varies by city|publisher=[[NBC News]]|date=12 April 2006|url=https://www.nbcnews.com/id/12287818|access-date=7 January 2007}} AP story concerning {{cite journal | pmid = 16611965 | doi=10.1056/NEJMc052959 | volume=354 | title=High levels of industrially produced trans fat in popular fast foods | date=April 2006 | journal=N. Engl. J. Med. | pages=1650–2 | last1 = Stender | first1 = S | last2 = Dyerberg | first2 = J | last3 = Astrup | first3 = A}}</ref> |
|||
===Nutritional guidelines=== |
|||
The [[United States National Academy of Sciences|National Academy of Sciences]] (NAS) advises the United States and Canadian governments on nutritional science for use in public policy and product labeling programs. Their 2002 ''Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids''<ref>{{cite book|last=Food and nutrition board, institute of medicine of the national academies|title=Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients)|publisher=National Academies Press|year=2005|pages=i|url=http://darwin.nap.edu/books/0309085373/html/R1.html|url-status=dead|archive-url=https://web.archive.org/web/20060918061042/http://darwin.nap.edu/books/0309085373/html/R1.html|archive-date=18 September 2006}}</ref> contains their findings and recommendations regarding consumption of trans fat ([http://www.nap.edu/openbook/0309085373/html/423.html summary]). |
|||
Their recommendations are based on two key facts. First, "trans fatty acids are not essential and provide no known benefit to human health",<ref name=p423/> whether of animal or plant origin.<ref>{{cite book|last=Food and nutrition board, institute of medicine of the national academies|title=Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients)|publisher=National Academies Press|year=2005|pages = 447|url=http://darwin.nap.edu/books/0309085373/html/447.html}}{{dead link|date=January 2018 |bot=InternetArchiveBot |fix-attempted=yes}}</ref> Second, while both saturated and trans fats increase levels of [[Low-density lipoprotein|LDL]], trans fats also lower levels of [[High-density lipoprotein|HDL]];<ref name=p504>{{cite book|last=Food and nutrition board, institute of medicine of the national academies|title=Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients)|publisher=National Academies Press|year=2005|pages = 504|url=http://darwin.nap.edu/books/0309085373/html/504.html}}{{dead link|date=September 2017 |bot=InternetArchiveBot |fix-attempted=yes}}</ref> thus increasing the risk of coronary artery disease. The NAS is concerned "that dietary trans fatty acids are more deleterious with respect to coronary artery disease than saturated fatty acids".<ref name=p504/> This analysis is supported by a 2006 [[New England Journal of Medicine]] (NEJM) scientific review that states "from a nutritional standpoint, the consumption of trans fatty acids results in considerable potential harm but no apparent benefit."<ref name="pmid16611951">{{cite journal | vauthors = Mozaffarian D, Katan MB, Ascherio A, Stampfer MJ, Willett WC | title = Trans fatty acids and cardiovascular disease | journal = The New England Journal of Medicine | volume = 354 | issue = 15 | pages = 1601–13 | date = April 2006 | pmid = 16611951 | doi = 10.1056/NEJMra054035 | url = https://semanticscholar.org/paper/e7d0b39670064e2b9b67ff88a8d243fa919ad9de }}</ref> |
|||
Because of these facts and concerns, the NAS has concluded there is no safe level of trans fat consumption. There is no adequate level, recommended daily amount or tolerable upper limit for trans fats. This is because any incremental increase in trans fat intake increases the risk of coronary artery disease.<ref name=p504/> |
|||
Despite this concern, the NAS dietary recommendations have not included eliminating trans fat from the diet. This is because trans fat is naturally present in many animal foods in trace quantities, and thus its removal from ordinary diets might introduce undesirable side effects and nutritional imbalances if proper nutritional planning is not undertaken. The NAS has, thus, "recommended that trans fatty acid consumption be as low as possible while consuming a nutritionally adequate diet".<ref>{{cite book|last=Food and nutrition board, institute of medicine of the national academies|title=Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients)|publisher=National Academies Press|year=2005|pages = 424|url=http://darwin.nap.edu/books/0309085373/html/424.html}}{{dead link|date=January 2018 |bot=InternetArchiveBot |fix-attempted=yes}}</ref> Like the NAS, the [[World Health Organization]] has tried to balance public health goals with a practical level of trans fat consumption, recommending in 2003 that trans fats be limited to less than 1% of overall energy intake.<ref name=Canada/> |
|||
The US National Dairy Council has asserted that the trans fats present in animal foods are of a different type than those in partially hydrogenated oils, and do not appear to exhibit the same negative effects.<ref name=dairy>{{cite journal| author=National Dairy Council|title=comments on 'Docket No. 2003N-0076 Food Labeling: Trans Fatty Acids in Nutrition Labeling'|date=18 June 2004|url=https://www.fda.gov/ohrms/dockets/dailys/04/June04/062304/03N-0076-emc00228-01.pdf|access-date=7 January 2007}}</ref> While a recent scientific review agrees with the conclusion (stating that "the sum of the current evidence suggests that the Public health implications of consuming trans fats from ruminant products are relatively limited"), it cautions that this may be due to the low consumption of trans fats from animal sources compared to artificial ones.<ref name="pmid16611951"/> |
|||
More recent inquiry (independent of the dairy industry) has found in a 2008 Dutch meta-analysis that all trans fats, regardless of natural or artificial origin equally raise LDL and lower HDL levels.<ref>{{cite journal | vauthors = Brouwer IA, Wanders AJ, Katan MB | title = Effect of animal and industrial trans fatty acids on HDL and LDL cholesterol levels in humans--a quantitative review | journal = PLOS ONE | volume = 5 | issue = 3 | pages = e9434 | date = March 2010 | pmid = 20209147 | pmc = 2830458 | doi = 10.1371/journal.pone.0009434 | editor1-last = Reitsma | bibcode = 2010PLoSO...5.9434B | editor1-first = Pieter H }}</ref> Other studies though have shown different results when it comes to animal based trans fats like conjugated linoleic acid (CLA). Although CLA is known for its anticancer properties, researchers have also found that the cis-9, trans-11 form of CLA can reduce the risk for cardiovascular disease and help fight inflammation.<ref name="Tricon S, Burdge GC, Kew S, et al. 2004 614–20">{{cite journal | vauthors = Tricon S, Burdge GC, Kew S, Banerjee T, Russell JJ, Jones EL, Grimble RF, Williams CM, Yaqoob P, Calder PC | display-authors = 6 | title = Opposing effects of cis-9,trans-11 and trans-10,cis-12 conjugated linoleic acid on blood lipids in healthy humans | journal = The American Journal of Clinical Nutrition | volume = 80 | issue = 3 | pages = 614–20 | date = September 2004 | pmid = 15321800 | doi = 10.1093/ajcn/80.3.614 | doi-access = free }}</ref><ref>{{cite journal | vauthors = Zulet MA, Marti A, Parra MD, Martínez JA | title = Inflammation and conjugated linoleic acid: mechanisms of action and implications for human health | journal = Journal of Physiology and Biochemistry | volume = 61 | issue = 3 | pages = 483–94 | date = September 2005 | pmid = 16440602 | doi = 10.1007/BF03168454 }}</ref> |
|||
===Health risks=== |
|||
Partially hydrogenated vegetable oils have been an increasingly significant part of the human diet for about 100 years (in particular, since the later half of the 20th century and where more processed foods are consumed),<ref name="Alfonso Valenzuel, Nora Morgado 1999">{{cite journal | vauthors = Valenzuela A, Morgado N | title = Trans fatty acid isomers in human health and in the food industry | journal = Biological Research | volume = 32 | issue = 4 | pages = 273–87 | year = 1999 | pmid = 10983247 | doi = 10.4067/s0716-97601999000400007 | doi-access = free }}</ref> and some deleterious effects of trans fat consumption are scientifically accepted, forming the basis of the health guidelines discussed above. |
|||
The exact [[biochemistry|biochemical]] process by which trans fats produce specific health problems are a topic of continuing research. Intake of dietary trans fat perturbs the body's ability to metabolize [[essential fatty acid]]s (EFAs, including [[Omega-3 fatty acid|Omega-3]]) leading to changes in the phospholipid fatty acid composition of the arterial walls, thereby raising risk of coronary artery disease.<ref>{{cite journal | vauthors = Kummerow FA, Zhou Q, Mahfouz MM, Smiricky MR, Grieshop CM, Schaeffer DJ | title = Trans fatty acids in hydrogenated fat inhibited the synthesis of the polyunsaturated fatty acids in the phospholipid of arterial cells | journal = Life Sciences | volume = 74 | issue = 22 | pages = 2707–23 | date = April 2004 | pmid = 15043986 | doi = 10.1016/j.lfs.2003.10.013 }}</ref> |
|||
While the mechanisms through which trans fatty acids contribute to coronary artery disease are fairly well understood, the mechanism for their effects on diabetes is still under investigation. They may impair the metabolism of long-chain polyunsaturated fatty acids (LCPUFAs),<ref>{{cite journal | vauthors = Mojska H | title = Influence of trans fatty acids on infant and fetus development | journal = Acta Microbiologica Polonica | volume = 52 Suppl | pages = 67–74 | year = 2003 | pmid = 15058815 }}</ref> but maternal pregnancy trans fatty acid intake has been inversely associated with LCPUFAs levels in infants at birth thought to underlie the positive association between breastfeeding and intelligence.<ref>{{cite journal | vauthors = Koletzko B, Decsi T | title = Metabolic aspects of trans fatty acids | journal = Clinical Nutrition | volume = 16 | issue = 5 | pages = 229–37 | date = October 1997 | pmid = 16844601 | doi = 10.1016/s0261-5614(97)80034-9 }}</ref> |
|||
High intake of trans fatty acids can lead to many health problems throughout one's life.<ref>{{cite journal | vauthors = Menaa F, Menaa A, Menaa B, Tréton J | title = Trans-fatty acids, dangerous bonds for health? A background review paper of their use, consumption, health implications and regulation in France | journal = European Journal of Nutrition | volume = 52 | issue = 4 | pages = 1289–302 | date = June 2013 | pmid = 23269652 | doi = 10.1007/s00394-012-0484-4 }}</ref> They are abundant in fast food restaurants.<ref name="Alfonso Valenzuel, Nora Morgado 1999"/> They are consumed in greater quantities by people who lack access to a diet consisting of fewer hydrogenated fats, or who often consume fast food. A diet high in trans fats can contribute to obesity, high blood pressure, and higher risk for heart disease{{citation needed|date=May 2014}}. Trans fat has also been implicated in the development of Type 2 diabetes.<ref>{{cite journal| first = Ulf | last = Riserus | name-list-format = vanc |title=Trans fatty acids, insulin sensitivity and type 2 diabetes|journal=Scandinavian Journal of Food and Nutrition|volume=50 |issue=4 |pages=161–165|year=2006|doi=10.1080/17482970601133114|doi-access=free}}</ref> |
|||
====Coronary artery disease==== |
|||
The primary health risk identified for trans fat consumption is an elevated risk of [[coronary artery disease]] (CAD).<ref name=TRANSformingApx9>{{cite journal|author=Trans Fat Task Force |title=TRANSforming the Food Supply (Appendix 9iii) |date=June 2006 |url=http://www.hc-sc.gc.ca/fn-an/nutrition/gras-trans-fats/tf-ge/tf-gt_app9iii_e.html |access-date=9 January 2007 |url-status=dead |archive-url=https://web.archive.org/web/20070225021532/http://www.hc-sc.gc.ca/fn-an/nutrition/gras-trans-fats/tf-ge/tf-gt_app9iii_e.html |archive-date=25 February 2007}} (Consultation on the health implications of alternatives to trans fatty acids: Summary of Responses from Experts)</ref> A 1994 study estimated that over 30,000 cardiac deaths per year in the United States are attributable to the consumption of trans fats.<ref name="pmid8179036" /> By 2006 upper estimates of 100,000 deaths were suggested.<ref name="B">{{cite journal | vauthors = Zaloga GP, Harvey KA, Stillwell W, Siddiqui R | title = Trans fatty acids and coronary heart disease | journal = Nutrition in Clinical Practice | volume = 21 | issue = 5 | pages = 505–12 | date = October 2006 | pmid = 16998148 | doi = 10.1177/0115426506021005505 }}</ref> A comprehensive review of studies of trans fats published in 2006 in the [[New England Journal of Medicine]] reports a strong and reliable connection between trans fat consumption and CAD, concluding that "On a per-calorie basis, trans fats appear to increase the risk of CAD more than any other macronutrient, conferring a substantially increased risk at low levels of consumption (1 to 3% of total energy intake)".<ref name="pmid16611951"/><ref name="pmid16611951"/> |
|||
The major evidence for the effect of trans fat on CAD comes from the [[Nurses' Health Study]] – a [[cohort study]] that has been following 120,000 female nurses since its inception in 1976. In this study, Hu and colleagues analyzed data from 900 coronary events from the study's [[statistical population|population]] during 14 years of followup. He determined that a nurse's CAD risk roughly doubled ([[relative risk]] of 1.93, [[confidence interval|CI]]: 1.43 to 2.61) for each 2% increase in trans fat calories consumed (instead of carbohydrate calories). By contrast, for each 5% increase in saturated fat calories (instead of carbohydrate calories) there was a 17% increase in risk ([[relative risk]] of 1.17, [[confidence interval|CI]]: 0.97 to 1.41). "The replacement of saturated fat or trans unsaturated fat by cis (unhydrogenated) unsaturated fats was associated with larger reductions in risk than an isocaloric replacement by carbohydrates."<ref name=nhs97>{{cite journal | vauthors = Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, Hennekens CH, Willett WC | display-authors = 6 | title = Dietary fat intake and the risk of coronary heart disease in women | journal = The New England Journal of Medicine | volume = 337 | issue = 21 | pages = 1491–9 | date = November 1997 | pmid = 9366580 | doi = 10.1056/NEJM199711203372102 }}</ref> Hu also reports on the benefits of reducing trans fat consumption. Replacing 2% of [[food energy]] from trans fat with non-trans [[unsaturated fat]]s more than halves the risk of CAD (53%). By comparison, replacing a larger 5% of food energy from saturated fat with non-trans unsaturated fats reduces the risk of CAD by 43%.<ref name=nhs97/> |
|||
Another study considered deaths due to CAD, with consumption of trans fats being linked to an increase in mortality, and consumption of [[polyunsaturated fat]]s being linked to a decrease in mortality.<ref name=TRANSformingApx9/><ref>{{cite journal | vauthors = Oh K, Hu FB, Manson JE, Stampfer MJ, Willett WC | title = Dietary fat intake and risk of coronary heart disease in women: 20 years of follow-up of the nurses' health study | journal = American Journal of Epidemiology | volume = 161 | issue = 7 | pages = 672–9 | date = April 2005 | pmid = 15781956 | doi = 10.1093/aje/kwi085 | doi-access = free }}</ref> |
|||
There are two accepted tests that measure an individual's risk for coronary artery disease, both [[blood test]]s. The first considers [[ratio]]s of two types of [[cholesterol]], the other the amount of a cell-signalling [[cytokine]] called [[C-reactive protein]]. The ratio test is more accepted, while the cytokine test may be more powerful but is still being studied.<ref name=TRANSformingApx9/> The effect of trans fat consumption has been documented on each as follows: |
|||
* Cholesterol ratio: This ratio compares the levels of [[low-density lipoprotein|LDL]] to [[High-density lipoprotein|HDL]]. Trans fat behaves like [[saturated fat]] by raising the level of LDL, but, unlike saturated fat, it has the additional effect of decreasing levels of HDL. The net increase in LDL/HDL ratio with trans fat is approximately double that due to saturated fat.<ref>{{cite journal | vauthors = Ascherio A, Katan MB, Zock PL, Stampfer MJ, Willett WC | title = Trans fatty acids and coronary heart disease | journal = The New England Journal of Medicine | volume = 340 | issue = 25 | pages = 1994–8 | date = June 1999 | pmid = 10379026 | doi = 10.1056/NEJM199906243402511 | url = https://semanticscholar.org/paper/901e1ee3995be35f030b958d93e3c9faa000c2a9 }}</ref><ref>{{cite journal | vauthors = Mensink RP, Katan MB | title = Effect of dietary trans fatty acids on high-density and low-density lipoprotein cholesterol levels in healthy subjects | journal = The New England Journal of Medicine | volume = 323 | issue = 7 | pages = 439–45 | date = August 1990 | pmid = 2374566 | doi = 10.1056/NEJM199008163230703 | url = http://library.wur.nl/WebQuery/wurpubs/13110 }}</ref><ref>{{cite journal | vauthors = Mensink RP, Zock PL, Kester AD, Katan MB | title = Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials | journal = The American Journal of Clinical Nutrition | volume = 77 | issue = 5 | pages = 1146–55 | date = May 2003 | pmid = 12716665 | doi = 10.1093/ajcn/77.5.1146 | doi-access = free }}</ref> (Higher ratios are worse.) One randomized [[crossover study]] published in 2003 comparing the effect [[postprandial|of eating a meal]] on blood lipids of (relatively) cis and trans fat rich meals showed that [[cholesteryl ester]] transfer (CET) was 28% higher after the trans meal than after the cis meal and that lipoprotein concentrations were enriched in [[apolipoprotein]](a) after the trans meals.<ref name="gatto">{{cite journal | vauthors = Gatto LM, Sullivan DR, Samman S | title = Postprandial effects of dietary trans fatty acids on apolipoprotein(a) and cholesteryl ester transfer | journal = The American Journal of Clinical Nutrition | volume = 77 | issue = 5 | pages = 1119–24 | date = May 2003 | pmid = 12716661 | doi = 10.1093/ajcn/77.5.1119 | doi-access = free }}</ref> |
|||
* C-reactive protein (CRP): A study of over 700 nurses showed that those in the highest [[quartile]] of trans fat consumption had blood levels of CRP that were 73% higher than those in the lowest quartile.<ref>{{cite journal | vauthors = Lopez-Garcia E, Schulze MB, Meigs JB, Manson JE, Rifai N, Stampfer MJ, Willett WC, Hu FB | display-authors = 6 | title = Consumption of trans fatty acids is related to plasma biomarkers of inflammation and endothelial dysfunction | journal = The Journal of Nutrition | volume = 135 | issue = 3 | pages = 562–6 | date = March 2005 | pmid = 15735094 | doi = 10.1093/jn/135.3.562 | doi-access = free }}</ref> |
|||
====Other health risks==== |
|||
There are suggestions that the negative consequences of trans fat consumption go beyond the cardiovascular risk. In general, there is much less scientific consensus asserting that eating trans fat specifically increases the risk of other chronic health problems: |
|||
* [[Alzheimer's Disease]]: A study published in [[Archives of Neurology]] in February 2003 suggested that the intake of both trans fats and saturated fats promote the development of Alzheimer disease,<ref>{{cite journal | vauthors = Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, Schneider J, Wilson RS | display-authors = 6 | title = Dietary fats and the risk of incident Alzheimer disease | journal = Archives of Neurology | volume = 60 | issue = 2 | pages = 194–200 | date = February 2003 | pmid = 12580703 | doi = 10.1001/archneur.60.2.194 | doi-access = free }}</ref> although not confirmed in an animal model.<ref name=phivilay /> It has been found that trans fats impaired memory and learning in middle-age rats. The trans-fat eating rats' brains had fewer proteins critical to healthy neurological function. Inflammation in and around the hippocampus, the part of the brain responsible for learning and memory. These are the exact types of changes normally seen at the onset of Alzheimer's, but seen after six weeks, even though the rats were still young.<ref>{{cite journal | vauthors = Granholm AC, Bimonte-Nelson HA, Moore AB, Nelson ME, Freeman LR, Sambamurti K | title = Effects of a saturated fat and high cholesterol diet on memory and hippocampal morphology in the middle-aged rat | journal = Journal of Alzheimer's Disease | volume = 14 | issue = 2 | pages = 133–45 | date = June 2008 | pmid = 18560126 | pmc = 2670571 | doi = 10.3233/JAD-2008-14202 }}</ref> |
|||
* [[Cancer]]: There is no scientific consensus that consuming trans fats significantly increases cancer risks across the board.<ref name=TRANSformingApx9/> The American Cancer Society states that a relationship between trans fats and cancer "has not been determined."<ref>{{cite web|last=American Cancer Society|title=Common questions about diet and cancer|url=http://www.cancer.org/docroot/ped/content/ped_3_2x_common_questions_about_diet_and_cancer.asp|access-date=9 January 2007}}</ref> One study has found a positive connection between trans fat and prostate cancer.<ref>{{cite journal| vauthors = Chavarro J, Stampfer M, Campos H, Kurth T, Willett W, Ma J |title=A prospective study of blood trans fatty acid levels and risk of prostate cancer|journal=Proc. Amer. Assoc. Cancer Res.|volume=47|date=1 April 2006|url=http://www.aacrmeetingabstracts.org/cgi/content/abstract/2006/1/943|access-date=9 January 2007|issue=1|pages = 943}}</ref> However, a larger study found a correlation between trans fats and a significant decrease in high-grade prostate cancer.<ref>{{cite journal | vauthors = Brasky TM, Till C, White E, Neuhouser ML, Song X, Goodman P, Thompson IM, King IB, Albanes D, Kristal AR | display-authors = 6 | title = Serum phospholipid fatty acids and prostate cancer risk: results from the prostate cancer prevention trial | journal = American Journal of Epidemiology | volume = 173 | issue = 12 | pages = 1429–39 | date = June 2011 | pmid = 21518693 | pmc = 3145396 | doi = 10.1093/aje/kwr027 }}</ref> An increased intake of trans fatty acids may raise the risk of breast cancer by 75%, suggest the results from the French part of the European Prospective Investigation into Cancer and Nutrition.<ref>{{cite press release|url=http://www.iarc.fr/ENG/Press_Releases/pr184a.html |title=Breast cancer: a role for trans fatty acids? |work=World Health Organization |date=11 April 2008 |url-status=dead |archive-url=https://web.archive.org/web/20080413023008/http://www.iarc.fr/ENG/Press_Releases/pr184a.html |archive-date=13 April 2008}}</ref><ref>{{cite journal | vauthors = Chajès V, Thiébaut AC, Rotival M, Gauthier E, Maillard V, Boutron-Ruault MC, Joulin V, Lenoir GM, Clavel-Chapelon F | display-authors = 6 | title = Association between serum trans-monounsaturated fatty acids and breast cancer risk in the E3N-EPIC Study | journal = American Journal of Epidemiology | volume = 167 | issue = 11 | pages = 1312–20 | date = June 2008 | pmid = 18390841 | pmc = 2679982 | doi = 10.1093/aje/kwn069 }}</ref> |
|||
* [[Diabetes]]: There is a growing concern that the risk of [[diabetes mellitus type 2|type 2 diabetes]] increases with trans fat consumption.<ref name=TRANSformingApx9/> However, consensus has not been reached.<ref name="pmid16611951"/> For example, one study found that risk is higher for those in the highest [[quartile]] of trans fat consumption.<ref>{{cite journal | vauthors = Hu FB, van Dam RM, Liu S | title = Diet and risk of Type II diabetes: the role of types of fat and carbohydrate | journal = Diabetologia | volume = 44 | issue = 7 | pages = 805–17 | date = July 2001 | pmid = 11508264 | doi = 10.1007/s001250100547 | doi-access = free }}</ref> Another study has found no diabetes risk once other factors such as total fat intake and BMI were accounted for.<ref>{{cite journal | vauthors = van Dam RM, Willett WC, Rimm EB, Stampfer MJ, Hu FB | title = Dietary fat and meat intake in relation to risk of type 2 diabetes in men | journal = Diabetes Care | volume = 25 | issue = 3 | pages = 417–24 | date = March 2002 | pmid = 11874924 | doi = 10.2337/diacare.25.3.417 | doi-access = free }}</ref> |
|||
* [[Obesity]]: Research indicates that trans fat may increase weight gain and abdominal fat, despite a similar caloric intake.<ref>{{cite news|last=Gosline|first=Anna | name-list-format = vanc |title=Why fast foods are bad, even in moderation|work=[[New Scientist]]|date=12 June 2006|url=https://www.newscientist.com/article.ns?id=dn9318|access-date=9 January 2007}}</ref> A 6-year experiment revealed that monkeys fed a trans fat diet gained 7.2% of their body weight, as compared to 1.8% for monkeys on a mono-unsaturated fat diet.<ref>{{cite journal |url= https://www.newscientist.com/channel/health/mg19025565.000-six-years-of-fastfood-fats-supersizes-monkeys.html |title=Six years of fast-food fats supersizes monkeys |journal=New Scientist |issue=2556 |date=17 June 2006 |pages = 21 }}</ref><ref name="Obesity 2007">{{cite journal | vauthors = Kavanagh K, Jones KL, Sawyer J, Kelley K, Carr JJ, Wagner JD, Rudel LL | title = Trans fat diet induces abdominal obesity and changes in insulin sensitivity in monkeys | journal = Obesity | volume = 15 | issue = 7 | pages = 1675–84 | date = July 2007 | pmid = 17636085 | doi = 10.1038/oby.2007.200 }}</ref> Although obesity is frequently linked to trans fat in the popular media,<ref>{{cite news |url=https://www.hhs.gov/news/speech/2003/030709.html |title=Trans Fat Press Conference | last = Thompson | first = Tommy G | name-list-format = vanc |url-status=dead |archive-url=https://web.archive.org/web/20060709233042/http://www.hhs.gov/news/speech/2003/030709.html |archive-date=9 July 2006 }}, US Secretary of health and human services</ref> this is generally in the context of eating too many calories; there is not a strong scientific consensus connecting trans fat and obesity, although the 6-year experiment did find such a link, concluding that "under controlled feeding conditions, long-term TFA consumption was an independent factor in weight gain. TFAs enhanced intra-abdominal deposition of fat, even in the absence of caloric excess, and were associated with insulin resistance, with evidence that there is impaired post-insulin receptor binding signal transduction."<ref name="Obesity 2007"/> |
|||
* [[Liver function|Liver dysfunction]]: Trans fats are metabolized differently by the [[liver]] than other fats and interfere with delta 6 [[desaturase]]. Delta 6 desaturase is an [[enzyme]] involved in converting essential fatty acids to [[arachidonic acid]] and [[prostaglandin]]s, both of which are important to the functioning of cells.<ref>{{cite journal | vauthors = Mahfouz M | title = Effect of dietary trans fatty acids on the delta 5, delta 6 and delta 9 desaturases of rat liver microsomes in vivo | journal = Acta Biologica et Medica Germanica | volume = 40 | issue = 12 | pages = 1699–1705 | year = 1981 | pmid = 7345825 }}</ref> |
|||
* [[Female infertility|Infertility in women]]: One 2007 study found, "Each 2% increase in the intake of energy from trans unsaturated fats, as opposed to that from carbohydrates, was associated with a 73% greater risk of ovulatory infertility...".<ref>{{cite journal | vauthors = Chavarro JE, Rich-Edwards JW, Rosner BA, Willett WC | title = Dietary fatty acid intakes and the risk of ovulatory infertility | journal = The American Journal of Clinical Nutrition | volume = 85 | issue = 1 | pages = 231–7 | date = January 2007 | pmid = 17209201 | doi = 10.1093/ajcn/85.1.231 | doi-access = free }}</ref> |
|||
* [[Major depressive disorder]]: Spanish researchers analysed the diets of 12,059 people over six years and found that those who ate the most trans fats had a 48 per cent higher risk of depression than those who did not eat trans fats.<ref>{{cite news|title=Trans fats and saturated fats could contribute to depression|url=https://www.smh.com.au/lifestyle/wellbeing/food-with-bad-fats-linked-to-depression-study-finds-20110127-1a6vy.html|work=The Sydney Morning Herald|date=28 January 2011|access-date=8 February 2011|first=Shari|last=Roan | name-list-format = vanc }}</ref> One mechanism may be trans-fats' substitution for [[docosahexaenoic acid]] (DHA) levels in the [[orbitofrontal cortex]] (OFC). Very high intake of trans-fatty acids (43% of total fat) in mice from 2 to 16 months of age was associated with lowered DHA levels in the brain (p=0.001).<ref name=phivilay>{{cite journal | vauthors = Phivilay A, Julien C, Tremblay C, Berthiaume L, Julien P, Giguère Y, Calon F | title = High dietary consumption of trans fatty acids decreases brain docosahexaenoic acid but does not alter amyloid-beta and tau pathologies in the 3xTg-AD model of Alzheimer's disease | journal = Neuroscience | volume = 159 | issue = 1 | pages = 296–307 | date = March 2009 | pmid = 19135506 | doi = 10.1016/j.neuroscience.2008.12.006 }}</ref> When the brains of 15 major depressive subjects who had committed suicide were examined post-mortem and compared against 27 age-matched controls, the suicidal brains were found to have 16% less (male average) to 32% less (female average) DHA in the OFC. The OFC controls [[Reward (psychology)|reward]], reward expectation, and [[empathy]] (all of which are reduced in depressive mood disorders) and regulates the [[limbic system]].<ref name=mcnamara>{{cite journal | vauthors = McNamara RK, Hahn CG, Jandacek R, Rider T, Tso P, Stanford KE, Richtand NM | title = Selective deficits in the omega-3 fatty acid docosahexaenoic acid in the postmortem orbitofrontal cortex of patients with major depressive disorder | journal = Biological Psychiatry | volume = 62 | issue = 1 | pages = 17–24 | date = July 2007 | pmid = 17188654 | doi = 10.1016/j.biopsych.2006.08.026 | url = http://www.biologicalpsychiatryjournal.com/article/S0006-3223(06)01065-1/abstract }}</ref> |
|||
* Behavioral [[irritability]] and [[aggression]]: a 2012 observational analysis of subjects of an earlier study found a strong relation between dietary trans fat acids and self-reported behavioral aggression and irritability, suggesting but not establishing causality.<ref>{{cite journal | vauthors = Golomb BA, Evans MA, White HL, Dimsdale JE | title = Trans fat consumption and aggression | journal = PLOS ONE | volume = 7 | issue = 3 | pages = e32175 | year = 2012 | pmid = 22403632 | pmc = 3293881 | doi = 10.1371/journal.pone.0032175 | bibcode = 2012PLoSO...732175G }}</ref> |
|||
* Diminished [[memory]]: In a 2015 article, researchers re-analyzing results from the 1999-2005 UCSD Statin Study argue that "greater dietary trans fatty acid consumption is linked to worse word memory in adults during years of high productivity, adults age <45".<ref>{{cite journal | vauthors = Golomb BA, Bui AK | title = A Fat to Forget: Trans Fat Consumption and Memory | journal = PLOS ONE | volume = 10 | issue = 6 | pages = e0128129 | year = 2015 | pmid = 26083739 | pmc = 4470692 | doi = 10.1371/journal.pone.0128129 | bibcode = 2015PLoSO..1028129G }}</ref> |
|||
* [[Acne]]: According to a 2015 study, trans fats are one of several components of [[Western pattern diet]]s which promote acne, along with [[carbohydrate]]s with high [[glycemic load]] such as [[white sugar|refined sugars]] or [[refined grains|refined starches]], [[milk]] and [[dairy product]]s, and [[saturated fat]]s, while [[omega-3 fatty acid]]s, which reduce acne, are deficient in Western pattern diets.<ref>{{cite journal | vauthors = Melnik BC | title = Linking diet to acne metabolomics, inflammation, and comedogenesis: an update | journal = Clinical, Cosmetic and Investigational Dermatology | volume = 8 | pages = 371–88 | date = 15 July 2015 | pmid = 26203267 | pmc = 4507494 | doi = 10.2147/CCID.S69135 | editor1-last = Weinberg | editor1-first = Jeffrey }}</ref> |
|||
===Public response and regulation=== |
|||
{{main|Trans fat regulation}} |
|||
== See also == |
|||
=== Further reading === |
|||
{{refbegin}} |
|||
* {{cite book |editor1-last=Dijkstra |editor1-first=Albert |editor2-last=Hamilton |editor2-first=Richard J. |editor3-last=Wolf |editor3-first=Hamm | name-list-format = vanc |title=Trans Fatty Acids |publisher=Blackwell |year=2008 |isbn=978-1-4051-5691-2 }} |
|||
* {{cite journal|url=http://members.ift.org/NR/rdonlyres/27B49B9B-EA63-4D73-BAB4-42FEFCD72C68/0/crfsfsv4n1p00220030ms20040577.pdf|vauthors=Jang ES, Jung MY, Min DB|title=Hydrogenation for Low Trans and High Conjugated Fatty Acids|journal=Comprehensive Reviews in Food Science and Food Safety|volume=1|year=2005|url-status=dead|archive-url=https://web.archive.org/web/20081217185908/http://members.ift.org/NR/rdonlyres/27B49B9B-EA63-4D73-BAB4-42FEFCD72C68/0/crfsfsv4n1p00220030ms20040577.pdf|archive-date=17 December 2008}} |
|||
{{refend}} |
|||
=== External links === |
|||
* [http://www.voicemagazine.org/articles/articledisplay.php?ART=5145 "Ban the Trans: These Sorry Lipids Should Go Away"] |
|||
* [http://www.cspinet.org/transfat/ Center for Science in the Public Interest Trans Fat Page] |
|||
* [http://www.hsph.harvard.edu/nutritionsource/transfats.html Harvard School of Public Health webpage on trans-fat] |
|||
* {{cite web|url=https://www.fda.gov/food/guidanceregulation/guidancedocumentsregulatoryinformation/labelingnutrition/ucm053479.htm |title=Labeling & Nutrition – Guidance for Industry: ''Trans'' Fatty Acids in Nutrition Labeling, Nutrient Content Claims, Health Claims; Small Entity Compliance Guide |date=August 2003 |publisher=Center for Food Safety and Applied Nutrition |access-date=6 April 2014 |archive-url=https://web.archive.org/web/20131026212809/https://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/ucm053479.htm |archive-date=26 October 2013 |url-status=live }} |
|||
* [https://www.fda.gov/OHRMS/DOCKETS/98fr/03-17525.htm Federal Register – 68 FR 41433 11 July 2003: Food Labeling: Trans Fatty Acids in Nutrition Labeling, Nutrient Content Claims, and Health Claims] |
|||
==Nutritional and health aspects== |
==Nutritional and health aspects== |
||
Revision as of 15:23, 30 August 2020

| Types of fats in food |
|---|
| Components |
| Manufactured fats |
In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds; most commonly those that occur in living beings or in food.[1]
The term often refers specifically to triglycerides (triple esters of glycerol), that are the main components of vegetable oils and of fatty tissue in animals and humans;[2] or, even more narrowly, to triglycerides that are solid or semisolid at room temperature, thus excluding oils. The term may also be used more broadly as a synonym of lipid -- any substance of biological relevance, composed of carbon, hydrogen, or oxygen, that is insoluble in water but soluble in non-polar solvents.[1] In this sense, besides the triglycerides, the term would include several other types of compounds like mono- and diglycerides, phospholipids (such as lecithin), sterols (such as cholesterol), waxes (such as beeswax),[1] and free fatty acids, which are usually present in human diet in smaller amounts.[2]
Fats are one of the three main macronutrient groups in human diet, along with carbohydrates and proteins,[1][3] and the main components of common food products like milk, butter, tallow, lard, bacon, and cooking oils. They are a major and dense source of food energy for many animals and play important structural and metabolic functions, in most living beings, including energy storage, waterproofing, and thermal insulation.[4] The human body can produce the fat that it needs from other food ingredients, except for a few essential fatty acids that must be included in the diet. Dietary fats are also the carriers of some flavor and aroma ingredients and vitamins that are not water-soluble.[2]
Chemical structure
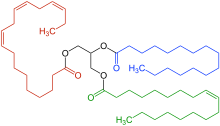
The most important elements in the chemical makeup of fats are the fatty acids. The molecule of a fatty acid consists of a carboxyl group HO(O=)C– connected to an unbranched alkyl group –(CH
x)
nH: namely, a chain of carbon atoms, joined by single, double, or (more rarely) triple bonds, with all remaining free bonds filled by hydrogen atoms[5]
The most common type of fat, in human diet and most living beings, is a triglyceride, an ester of the triple alcohol glycerol H(–CHOH–)
3H and three fatty acids. The molecule of a trigliceride can be described as resulting from a condensation reaction (specifically, esterification) between each of glycerol's –OH groups and the HO– part of the carboxyl group HO(O=)C– of each fatty acid, forming an ester bridge –O–(O=)C– with elimination of a water molecule H
2O.
Other less common types of fats include diglycerides and monoglycerides, where the esterification is limited to two or just one of glycerol's –OH groups. Other alcohols, such as cetyl alcohol (predominant in spermaceti), may replace glycerol. In the phospholipids, one of the fatty acids is replaced by phosphoric acid or a monoester thereof.
Conformation
The shape of fat and fatty acid molecules is usually not well-defined. Any two parts of a molecule that are connected by just one single bond are free to rotate about that bond. Thus a fatty acid molecule with n simple bonds can be deformed in n-1 independent ways (counting also rotation of the terminal methyl group).
Such rotation cannot happen across a double bond, except by breaking and then reforming it with one of the halves of the molecule rotated by 180 degrees, which requires crossing a significant energy barrier. Thus a fat or fatty acid molecule with double bonds (excluding at the very end of the chain) can have multiple cis-trans isomers with significantly different chemical and biological properties. Each double bond reduces the number of conformational degrees of freedom by one. Each triple bond forces the four nearest carbons to lie in a straight line, removing two degrees of freedom.
It follows that depictions of "saturated" fatty acids with no double bonds (like stearic) having a "straight zig-zag" shape, and those with one cis bond (like oleic) being bent in an "elbow" shape are somewhat misleading. While the latter are a little less flexible, both can be twisted to assume similar straight or elbow shapes. In fact, outside of some specific contxts like crystals or bilayer membranes, both are more likely to be found in randomly contorted configurations than in either of those two shapes.
Examples
| Stearic acid saturated |

|
|---|---|
| Oleic acid unsaturated cis-8) |

|
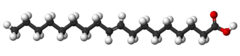
| Elaidic acid unsaturated trans-8) |
|
| Vaccenic acid unsaturated trans-11) |
|
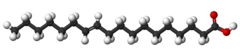
Stearic acid is a saturated fatty acid (with only single bonds) found in animal fats, and is the intended product in full hydrogenation.
Oleic acid has a double bond (thus being "unsaturated") with cis geometry about midway in the chain; it makes up 55–80% of olive oil.
Elaidic acid is its trans isomer; it may be present in partially hydrogenated vegetable oils, and also occurs in the fat of the durian fruit (about 2%) and in milk fat (less than 0.1%).
Vaccenic acid is another trans acid that differs from elaidic only in the position of the double bond; it also occurs in milk fat (about 1-2%).
Nomenclature
Common fat names
Fats are usually named after their source (like butterfat, olive oil, cod liver oil, tail fat) or have traditional names of their own (like butter, lard, ghee, and margarine). Some of these names refer to products that contain substantial amounts of other components besides fats proper.
Chemical fatty acid names
In chemistry and biochemistry, dozens of saturated fatty acids and of hundreds of unsaturated ones have proper scientfic/technical names usually inspired by their source fats (butyric, caprylic, stearic, oleic, palmitic, and nervonic), but sometimes their discoverer (mead, osbond).
A triglyceride would then be named as an ester of those acids, such as "glyceryl 1,2-dioleate 3-palmitate".[6]
IUPAC
In the general chemical nomenclature developed by the International Union of Pure and Applied Chemistry (IUPAC), the recommended name of a fatty acid, derived from the name of the corresponding hydrocarbon, completely describes its structure, by specifying the number of carbons and the number and position of the double bonds. Thus, for example, oleic acid would be called "(9Z)-octadec-9-enoic acid", meaning that it has a 18 carbon chain ("octadec") with a carboxyl at one end ("oic") and a double bound at carbon 9 counting from the carboxyl ("9-en"), and that the configuration of the single bonds adjacent to that double bond is cis ("(9Z)") The IUPAC nomenclature can also handle branched chains and derivatives where hydrogen atoms are replaced by other chemical groups.
A triglyceride would then be named according to general ester rules as, for example, "propane-1,2,3-tryl 1,2-bis((9Z)-octadec-9-enoate) 3-(hexadecanoate)".
Fatty acid code
A notation specific for fatty acids with unbranched chain, that is as precise as the IUPAC one but easier to parse, is a code of the form "{N}:{D} cis-{CCC} trans-{TTT}", where {N} is the number of carbons (including the carboxyl one), {D} is the number of double bonds, {CCC} is a list of the positions of the cis double bonds, and {TTT} is a list of the postions of the trans bounds. Either list and the label is omitted if there are no bounds of that type.
Thus, for example, the codes for stearic, oleic, elaidic, and vaccenic acids would be "18:0", "18:1 cis-9", "18:1 trans-9", and "18:1 trans-11", respectively. The code for α-oleostearic acid, which is "(9E,11E,13Z)-octadeca-9,11,13-trienoic acid" in the IUPAC nomenclature, has the code "18:3 trans-9,11 cis-13"
Classification
By chain length
Fats can be classified according to the lengths of the carbon chains of their constituent fatty acids. Most chemical properties, such as melting point and acidity, vary gradually with this parameter, so there is no sharp division. Chemically, formic acid (1 carbon) and acetic acid (2 carbons) could be viewed as the shortest fatty acids; then triformin would be the simplest trigliceride. However, the terms "fatty acid" and "fat" are usually reserved for compounds with substantially longer chains.[citation needed]
A division commonly made in biochemistry and nutrition is:[citation needed]
- Short-chain fatty acid (SCFA) with less than six carbons (e. g. butyric acid).
- Medium-chain fatty acid (MCFA) with 6 to 12 carbons (e.g. capric acid).
- Long-chain fatty acids (LCFA) with 13 to 21 carbons (e.g. petroselinic acid).
- Very long chain fatty acids (VLCFA) with 22 or more carbons (e. g. cerotic acid with 26)
A triglyceride molecule may have fatty acid elements of different lengths, and a fat product will often be a mix of various triglycerides. Most fats found in food, whether vegetable or animal, are made up of medium to long-chain fatty acids, usually of equal or nearly equal length.
Saturated and unsaturated fats
For human nutrition, an important classification of fats is based on the number and position of double bonds in the constituent fatty acids. Saturated fat has a predominance of saturated fatty acids, without any double bonds, while unsaturated fat has predominantly unsaturated acids with double bonds.
Unsaturated fatty acids are further classified into "monounsaturated", with a single double bond, and "polyunsaturated" with two or more. Natural fats usually contain several different saturated and unsaturated acids, even on the same molecule. For example, in most vegetable oils, the saturated palmitic (C16:0) and stearic (C18:0) acid residues are usually attached to positions 1 and 3 (sn1 and sn3) of the glycerol hub, whereas the middle position (sn2) is usually occupied by an unsaturated one, such as oleic (C18:1, ω–9) or linoleic (C18:2, ω–6).[7])
Saturated fats generally have a higher melting point than usaturated ones with the same molecular weight, and thus are more likely to be solid at room temperature. For example, the animal fats tallow and lard are high in saturated fatty acid content and are solids. Olive and linseed oils on the other hand are unsaturated and liquid. Unsaturated fats are prone to oxidation by air, which causes them to become rancid and inedible.
The double bonds in unsaturated fats can be converted into single bonds by reaction with hydrogen effected by a catalyst. This process, called hydrogenation, is used to turn vegetable vegetable oils into solid or semisolid vegetable fats like margarine, which can substitute for tallow and butter and (unlike unsaturated fats) can be stored indefinitely without becoming rancid. However, partial hydrogenation also creates some unwanted trans acids from cis acids.[citation needed]
Cis and trans fats
Another important classification of unsaturated fatty acids considers the cis-trans isomerism, the spatial arrangement of the single bonds adjacent to the double bonds. Most unsaturated fatty acids that occur in nature have those bonds in the cis ("same side") configuration.
Omega number
Another classification considers the position of double bonds, counted from the "ω" (omega) or "n" carbon atom at the end opposite to the carboxyl group. Thus, for example, alpha-linolenic acid is an omega-3 fatty acid because the 3rd carbon from that end is the first to have a double bond.
Biological importance
In humans and many animals, fats serve both as energy sources and as stores for energy in excess of what the body needs immediately. Each gram of fat when burned or metabolized releases about 9 food calories (37 kJ = 8.8 kcal).[8]
Fats are also sources of essential fatty acids, an important dietary requirement. Vitamins A, D, E, and K are fat-soluble, meaning they can only be digested, absorbed, and transported in conjunction with fats.
Fats play a vital role in maintaining healthy skin and hair, insulating body organs against shock, maintaining body temperature, and promoting healthy cell function. Fat also serves as a useful buffer against a host of diseases. When a particular substance, whether chemical or biotic, reaches unsafe levels in the bloodstream, the body can effectively dilute—or at least maintain equilibrium of—the offending substances by storing it in new fat tissue.[citation needed] This helps to protect vital organs, until such time as the offending substances can be metabolized or removed from the body by such means as excretion, urination, accidental or intentional bloodletting, sebum excretion, and hair growth.
Adipose tissue
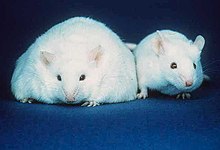
In animals, adipose tissue, or fatty tissue is the body's means of storing metabolic energy over extended periods of time. Adipocytes (fat cells) store fat derived from the diet and from liver metabolism. Under energy stress these cells may degrade their stored fat to supply fatty acids and also glycerol to the circulation. These metabolic activities are regulated by several hormones (e.g., insulin, glucagon and epinephrine). Adipose tissue also secretes the hormone leptin.[9]
The location of the tissue determines its metabolic profile: visceral fat is located within the abdominal wall (i.e., beneath the wall of abdominal muscle) whereas subcutaneous fat is located beneath the skin (and includes fat that is located in the abdominal area beneath the skin but above the abdominal muscle wall). Visceral fat was recently discovered to be a significant producer of signaling chemicals (i.e., hormones), among which several are involved in inflammatory tissue responses. One of these is resistin which has been linked to obesity, insulin resistance, and Type 2 diabetes. This latter result is currently controversial, and there have been reputable studies supporting all sides on the issue.[citation needed]
Production and processing
A variety of chemical and physical techniques are used for the production and processing of fats, both industrially and in cottage or home settings. They include:
- Pressing to extract liquid fats from fruits, seeds, or algae, e.g. olive oil from olives;
- Solvent extraction using solvents like hexane or supercritical carbon dioxide.
- Rendering, the melting of fat in adipose tissue, e.g. to produce tallow, lard, fish oil, and whale oil.
- Churning of milk to produce butter.
- Hydrogenation to reduce the degree of unsaturation of the fatty acids.
- Interesterification, the rearrangement of fatty acids across different triglicerides.
- Winterization to remove oil components with higher melting points.
- Clarification of butter.
Saturated fat
A saturated fat is a type of fat in which the fatty acid chains have all or predominantly single bonds. A fat is made of two kinds of smaller molecules: glycerol and fatty acids. Fats are made of long chains of carbon (C) atoms. Some carbon atoms are linked by single bonds (-C-C-) and others are linked by double bonds (-C=C-).[10] Double bonds can react with hydrogen to form single bonds. They are called saturated because the second bond is broken and each half of the bond is attached to (saturated with) a hydrogen atom.
Saturated fats tend to have higher melting points than their corresponding unsaturated fats, leading to the popular understanding that saturated fats tend to be solids at room temperatures, while unsaturated fats tend to be liquid at room temperature with varying degrees of viscosity.
Most animal fats are saturated. The fats of plants and fish are generally unsaturated.[10] Various foods contain different proportions of saturated and unsaturated fat. Many processed foods like foods deep-fried in hydrogenated oil and sausage are high in saturated fat content. Some store-bought baked goods are as well, especially those containing partially hydrogenated oils.[11][12][13] Other examples of foods containing a high proportion of saturated fat and dietary cholesterol include animal fat products such as lard or schmaltz, fatty meats and dairy products made with whole or reduced fat milk like yogurt, ice cream, cheese and butter.[14] Certain vegetable products have high saturated fat content, such as coconut oil and palm kernel oil.[15]
Guidelines released by many medical organizations, including the World Health Organization, have advocated for reduction in the intake of saturated fat to promote health and reduce the risk from cardiovascular diseases. Many review articles also recommend a diet low in saturated fat and argue it will lower risks of cardiovascular diseases,[16] diabetes, or death.[17] A small number of contemporary reviews have challenged these conclusions, though predominant medical opinion is that saturated fat and cardiovascular disease are closely related.[18][19][20]
Fat profiles
While nutrition labels regularly combine them, the saturated fatty acids appear in different proportions among food groups. Lauric and myristic acids are most commonly found in "tropical" oils (e.g., palm kernel, coconut) and dairy products. The saturated fat in meat, eggs, cacao, and nuts is primarily the triglycerides of palmitic and stearic acids.
| Food | Lauric acid | Myristic acid | Palmitic acid | Stearic acid |
|---|---|---|---|---|
| Coconut oil | 47% | 18% | 9% | 3% |
| Palm kernel oil | 48% | 1% | 44% | 5% |
| Butter | 3% | 11% | 29% | 13% |
| Ground beef | 0% | 4% | 26% | 15% |
| Salmon | 0% | 1% | 29% | 3% |
| Egg yolks | 0% | 0.3% | 27% | 10% |
| Cashews | 2% | 1% | 10% | 7% |
| Soybean oil | 0% | 0% | 11% | 4% |
Examples of saturated fatty acids
Some common examples of fatty acids:
- Butyric acid with 4 carbon atoms (contained in butter)
- Lauric acid with 12 carbon atoms (contained in coconut oil, palm kernel oil, and breast milk)
- Myristic acid with 14 carbon atoms (contained in cow's milk and dairy products)
- Palmitic acid with 16 carbon atoms (contained in palm oil and meat)
- Stearic acid with 18 carbon atoms (also contained in meat and cocoa butter)
Association with diseases
Cardiovascular disease
The effect of saturated fat on heart disease has been extensively studied.[22] There are strong, consistent, and graded relationships between saturated fat intake, blood cholesterol levels, and the epidemic of cardiovascular disease.[17] The relationships are accepted as causal.[23][24]
Many health authorities such as the Academy of Nutrition and Dietetics,[25] the British Dietetic Association,[26] American Heart Association,[17] the World Heart Federation,[27] the British National Health Service,[28] among others,[29][30] advise that saturated fat is a risk factor for cardiovascular disease. The World Health Organization in May 2015 recommends switching from saturated to unsaturated fats.[31]
There is moderate quality evidence that reducing the proportion of saturated fat in the diet, and replacing it with unsaturated fats or carbohydrates over a period of at least two years, leads to a reduction in the risk of cardiovascular disease.[22]
Dyslipidemia
The consumption of saturated fat is generally considered a risk factor for dyslipidemia, which in turn is a risk factor for some types of cardiovascular disease.[32][33][34][35][36]
Abnormal blood lipid levels, that is high total cholesterol, high levels of triglycerides, high levels of low-density lipoprotein (LDL, "bad" cholesterol) or low levels of high-density lipoprotein (HDL, "good" cholesterol) cholesterol are all associated with increased risk of heart disease and stroke.[27]
Meta-analyses have found a significant relationship between saturated fat and serum cholesterol levels.[17][37] High total cholesterol levels, which may be caused by many factors, are associated with an increased risk of cardiovascular disease.[38][39] However, other indicators measuring cholesterol such as high total/HDL cholesterol ratio are more predictive than total serum cholesterol.[39] In a study of myocardial infarction in 52 countries, the ApoB/ApoA1 (related to LDL and HDL, respectively) ratio was the strongest predictor of CVD among all risk factors.[40] There are other pathways involving obesity, triglyceride levels, insulin sensitivity, endothelial function, and thrombogenicity, among others, that play a role in CVD, although it seems, in the absence of an adverse blood lipid profile, the other known risk factors have only a weak atherogenic effect.[41] Different saturated fatty acids have differing effects on various lipid levels.[42]
Cancer
Breast cancer
A meta-analysis published in 2003 found a significant positive relationship in both control and cohort studies between saturated fat and breast cancer.[43] However two subsequent reviews have found weak or insignificant associations of saturated fat intake and breast cancer risk,[44][45] and note the prevalence of confounding factors.[44][46]
Colorectal cancer
One review found limited evidence for a positive relationship between consuming animal fat and incidence of colorectal cancer.[47]
Ovarian cancer
Meta-analyses of clinical studies found evidence for increased risk of ovarian cancer by high consumption of saturated fat.[48]
Prostate cancer
Some researchers have indicated that serum myristic acid[49][50] and palmitic acid[50] and dietary myristic[51] and palmitic[51] saturated fatty acids and serum palmitic combined with alpha-tocopherol supplementation[49] are associated with increased risk of prostate cancer in a dose-dependent manner. These associations may, however, reflect differences in intake or metabolism of these fatty acids between the precancer cases and controls, rather than being an actual cause.[50]
Bones
Mounting evidence indicates that the amount and type of fat in the diet can have important effects on bone health. Most of this evidence is derived from animal studies. The data from one study indicated that bone mineral density is negatively associated with saturated fat intake, and that men may be particularly vulnerable.[52]
Dietary recommendations
Recommendations to reduce or limit dietary intake of saturated fats are made by the World Health Organization,[53] American Heart Association,[17] Health Canada,[54] the US Department of Health and Human Services,[55] the UK National Health Service,[56] the Australian Department of Health and Aging,[57] the Singapore Ministry of Health,[58] the Indian Ministry of Health and Family Wellfare,[59] the New Zealand Ministry of Health,[60] and Hong Kong's Department of Health.[61]
In 2003, the World Health Organization (WHO) and Food and Agriculture Organization (FAO) expert consultation report concluded that "intake of saturated fatty acids is directly related to cardiovascular risk.[62] The traditional target is to restrict the intake of saturated fatty acids to less than 10% of daily energy intake and less than 7% for high-risk groups. If populations are consuming less than 10%, they should not increase that level of intake. Within these limits, intake of foods rich in myristic and palmitic acids should be replaced by fats with a lower content of these particular fatty acids. In developing countries, however, where energy intake for some population groups may be inadequate, energy expenditure is high and body fat stores are low (BMI <18.5 kg/m2). The amount and quality of fat supply has to be considered keeping in mind the need to meet energy requirements. Specific sources of saturated fat, such as coconut and palm oil, provide low-cost energy and may be an important source of energy for the poor."[62]
A 2004 statement released by the Centers for Disease Control (CDC) determined that "Americans need to continue working to reduce saturated fat intake…"[63] In addition, reviews by the American Heart Association led the Association to recommend reducing saturated fat intake to less than 7% of total calories according to its 2006 recommendations.[64][65] This concurs with similar conclusions made by the US Department of Health and Human Services, which determined that reduction in saturated fat consumption would positively affect health and reduce the prevalence of heart disease.[66]
The United Kingdom, National Health Service claims the majority of British people eat too much saturated fat. The British Heart Foundation also advises people to cut down on saturated fat. People are advised to cut down on saturated fat and read labels on food they buy.[67][68]
A 2004 review stated that "no lower safe limit of specific saturated fatty acid intakes has been identified" and recommended that the influence of varying saturated fatty acid intakes against a background of different individual lifestyles and genetic backgrounds should be the focus in future studies.[69]
Blanket recommendations to lower saturated fat were criticized at a 2010 conference debate of the American Dietetic Association for focusing too narrowly on reducing saturated fats rather than emphasizing increased consumption of healthy fats and unrefined carbohydrates. Concern was expressed over the health risks of replacing saturated fats in the diet with refined carbohydrates, which carry a high risk of obesity and heart disease, particularly at the expense of polyunsaturated fats which may have health benefits. None of the panelists recommended heavy consumption of saturated fats, emphasizing instead the importance of overall dietary quality to cardiovascular health.[70]
In a 2017 comprehensive review of the literature and clinical trials, the American Heart Association published a recommendation that saturated fat intake be reduced or replaced by products containing monounsaturated and polyunsaturated fats, a dietary adjustment that could reduce the risk of cardiovascular diseases by 30%.[17]
Molecular description


The two-dimensional illustration has implicit hydrogen atoms bonded to each of the carbon atoms in the polycarbon tail of the myristic acid molecule (there are 13 carbon atoms in the tail; 14 carbon atoms in the entire molecule).
Carbon atoms are also implicitly drawn, as they are portrayed as intersections between two straight lines. "Saturated," in general, refers to a maximum number of hydrogen atoms bonded to each carbon of the polycarbon tail as allowed by the Octet Rule. This also means that only single bonds (sigma bonds) will be present between adjacent carbon atoms of the tail.
Unsaturated fat
An unsaturated fat is a fat or fatty acid in which there is at least one double bond within the fatty acid chain. A fatty acid chain is monounsaturated if it contains one double bond, and polyunsaturated if it contains more than one double bond.
Where double bonds are formed, hydrogen atoms are subtracted from the carbon chain. Thus, a saturated fat has no double bonds, has the maximum number of hydrogens bonded to the carbons, and therefore is "saturated" with hydrogen atoms. In cellular metabolism, unsaturated fat molecules contain somewhat less energy (i.e., fewer calories) than an equivalent amount of saturated fat. The greater the degree of unsaturation in a fatty acid (i.e., the more double bonds in the fatty acid) the more vulnerable it is to lipid peroxidation (rancidity). Antioxidants can protect unsaturated fat from lipid peroxidation.
Chemistry and nutrition

Double bonds may be in either a cis or a trans isomer, depending on the geometry of the double bond. In the cis isomer, hydrogen atoms are on the same side of the double bond; whereas in the trans isomer, they are on opposite sides of the double bond (see trans fat). Saturated fats are useful in processed foods because saturated fats are less vulnerable to rancidity and usually more solid at room temperature than unsaturated fats. Unsaturated chains have a lower melting point, hence these molecules increase the fluidity of cell membranes.
Although both monounsaturated and polyunsaturated fats can replace saturated fat in the diet, trans unsaturated fats should not. Replacing saturated fats with unsaturated fats helps lower levels of total cholesterol and LDL cholesterol in the blood.[71] Trans unsaturated fats are an exception because the double bond stereochemistry predisposes the carbon chains to assume a linear conformation, which conforms to rigid packing as in plaque formation. The geometry of the cis double bond induces a bend in the molecule, thereby precluding rigid formations (see Trans fat § Chemistry links above for drawings that illustrate this). Natural sources of fatty acids (see above) are rich in the cis isomer[citation needed].
Although polyunsaturated fats are protective against cardiac arrhythmias, a study of post-menopauseal women with a relatively low fat intake showed that polyunsaturated fat is positively associated with progression of coronary atherosclerosis, whereas monounsaturated fat is not.[72] This probably is an indication of the greater vulnerability of polyunsaturated fats to lipid peroxidation, against which vitamin E has been shown to be protective.[73]
Examples of unsaturated fatty acids are palmitoleic acid, oleic acid, myristoleic acid, linoleic acid, and arachidonic acid. Foods containing unsaturated fats include avocado, nuts, olive oils, and vegetable oils such as canola. Meat products contain both saturated and unsaturated fats.
Although unsaturated fats are conventionally regarded as 'healthier' than saturated fats,[74] the United States Food and Drug Administration (FDA) recommendation stated that the amount of unsaturated fat consumed should not exceed 30% of one's daily caloric intake.[citation needed] Most foods contain both unsaturated and saturated fats. Marketers advertise only one or the other, depending on which one makes up the majority. Thus, various unsaturated fat vegetable oils, such as olive oils, also contain saturated fat.[75]
In chemical analysis, fatty acids are separated by gas chromatography of methyl esters;[76] additionally, a separation of unsaturated isomers is possible by argentation thin-layer chromatography.[77]
Role of dietary fats in insulin resistance
Incidence of insulin resistance is lowered with diets higher in monounsaturated fats (especially oleic acid), while the opposite is true for diets high in polyunsaturated fats (especially large amounts of arachidonic acid) as well as saturated fats (such as arachidic acid). These ratios can be indexed in the phospholipids of human skeletal muscle and in other tissues as well. This relationship between dietary fats and insulin resistance is presumed secondary to the relationship between insulin resistance and inflammation, which is partially modulated by dietary fat ratios (Omega-3/6/9) with both omega 3 and 9 thought to be anti-inflammatory, and omega 6 pro-inflammatory (as well as by numerous other dietary components, particularly polyphenols and exercise, with both of these anti-inflammatory). Although both pro- and anti-inflammatory types of fat are biologically necessary, fat dietary ratios in most US diets are skewed towards Omega 6, with subsequent disinhibition of inflammation and potentiation of insulin resistance.[75] But this is contrary to the suggestion of more recent studies, in which polyunsaturated fats are shown as protective against insulin resistance.
Membrane composition as a metabolic pacemaker
Studies on the cell membranes of mammals and reptiles discovered that mammalian cell membranes are composed of a higher proportion of polyunsaturated fatty acids (DHA, omega-3 fatty acid) than reptiles.[89] Studies on bird fatty acid composition have noted similar proportions to mammals but with 1/3rd less omega-3 fatty acids as compared to omega-6 for a given body size.[90] This fatty acid composition results in a more fluid cell membrane but also one that is permeable to various ions (H+ & Na+), resulting in cell membranes that are more costly to maintain. This maintenance cost has been argued to be one of the key causes for the high metabolic rates and concomitant warm-bloodedness of mammals and birds.[89] However polyunsaturation of cell membranes may also occur in response to chronic cold temperatures as well. In fish increasingly cold environments lead to increasingly high cell membrane content of both monounsaturated and polyunsaturated fatty acids, to maintain greater membrane fluidity (and functionality) at the lower temperatures.[91][92]
Monounsaturated fat
In biochemistry and nutrition, monounsaturated fatty acids (abbreviated MUFAs, or more plainly monounsaturated fats) are fatty acids that have one double bond in the fatty acid chain with all of the remainder carbon atoms being single-bonded. By contrast, polyunsaturated fatty acids (PUFAs) have more than one double bond.
Molecular description
Fatty acids are long-chained molecules having an alkyl group at one end and a carboxylic acid group at the other end. Fatty acid viscosity (thickness) and melting temperature increases with decreasing number of double bonds; therefore, monounsaturated fatty acids have a higher melting point than polyunsaturated fatty acids (more double bonds) and a lower melting point than saturated fatty acids (no double bonds). Monounsaturated fatty acids are liquids at room temperature and semisolid or solid when refrigerated resulting in an isotopic lattice structure.
Common monounsaturated fatty acids are palmitoleic acid (16:1 n−7), cis-vaccenic acid (18:1 n−7) and oleic acid (18:1 n−9). Palmitoleic acid has 16 carbon atoms with the first double bond occurring 7 carbon atoms away from the methyl group (and 9 carbons from the carboxyl end). It can be lengthened to the 18-carbon cis-vaccenic acid. Oleic acid has 18 carbon atoms with the first double bond occurring 9 carbon atoms away from the carboxylic acid group. The illustrations below show a molecule of oleic acid in Lewis formula and as a space-filling model.


List of monounsaturated fats
| Common name | Lipid name | Chemical name |
|---|---|---|
| Myristoleic acid | 14:1 (n-5) | cis-Tetradec-9-enoic acid |
| Palmitoleic acid | 16:1 (n-7) | cis-Hexadec-9-enoic acid |
| cis-Vaccenic acid | 18:1 (n-7) | cis-Octadec-11-enoic acid |
| Vaccenic acid | 18:1 (n-7) | trans-Octadec-11-enoic acid |
| Paullinic acid | 20:1 (n-7) | cis-13-Eicosenoic acid |
| Oleic acid | 18:1 (n-9) | cis-Octadec-9-enoic acid |
| Elaidic acid (trans-oleic acid) | 18:1 (n-9) | trans-Octadec-9-enoic acid |
| 11-Eicosenoic acid (gondoic acid) | 20:1 (n-9) | cis-Eicos-11-enoic acid |
| Erucic acid | 22:1 (n-9) | cis-Tetracos-15-enoic acid |
| Brassidic acid | 22:1 (n-9) | trans-Tetracos-15-enoic acid |
| Nervonic acid | 24:1 (n-9) | cis-Tetracos-15-enoic acid |
| Sapienic acid | 16:1 (n-10) | cis-6-Hexadecenoic acid |
| Gadoleic acid | 20:1 (n-11) | cis-9-Icosenoic acid |
| Petroselinic acid | 18:1 (n-12) | cis-Octadec-6-enoic acid |
Health
The large scale KANWU study found that increasing monounsaturated fat and decreasing saturated fat intake could improve insulin sensitivity, but only when the overall fat intake of the diet was low.[93] However, some monounsaturated fatty acids (in the same way as saturated fats) may promote insulin resistance, whereas polyunsaturated fatty acids may be protective against insulin resistance.[94][95] Studies have shown that substituting dietary monounsaturated fat for saturated fat is associated with increased daily physical activity and resting energy expenditure. More physical activity was associated with a higher-oleic acid diet than one of a palmitic acid diet. From the study, it is shown that more monounsaturated fats lead to less anger and irritability.[96]
Foods containing monounsaturated fats reduce low-density lipoprotein (LDL) cholesterol,[97] while possibly increasing high-density lipoprotein (HDL) cholesterol.[98]
Levels of oleic acid along with other monounsaturated fatty acids in red blood cell membranes were positively associated with breast cancer risk. The saturation index (SI) of the same membranes was inversely associated with breast cancer risk. Monounsaturated fats and low SI in erythrocyte membranes are predictors of postmenopausal breast cancer. Both of these variables depend on the activity of the enzyme delta-9 desaturase (Δ9-d).[99]
In children, consumption of monounsaturated oils is associated with healthier serum lipid profiles.[100]
The Mediterranean diet is one heavily influenced by monounsaturated fats. People in Mediterranean countries consume more total fat than Northern European countries, but most of the fat is in the form of monounsaturated fatty acids from olive oil and omega-3 fatty acids from fish, vegetables, and certain meats like lamb, while consumption of saturated fat is minimal in comparison. A 2017 review found evidence that the practice of a Mediterranean diet could lead to a decreased risk of cardiovascular diseases, overall cancer incidence, neurodegenerative diseases, diabetes, and early death.[101] A 2018 review showed that the practice of the Mediterranean diet may improve overall health status, such as reduced risk of non-communicable diseases. It also may reduce the social and economic costs of diet-related illnesses.[102]
Sources
Monounsaturated fats are found in animal flesh such as red meat, whole milk products, nuts, and high fat fruits such as olives and avocados. Algal oil is about 92% monounsaturated fat.[103] Olive oil is about 75% monounsaturated fat.[104] The high oleic variety sunflower oil contains at least 70% monounsaturated fat.[105] Canola oil and cashews are both about 58% monounsaturated fat.[citation needed] Tallow (beef fat) is about 50% monounsaturated fat.[106] and lard is about 40% monounsaturated fat.[citation needed] Other sources include hazelnut, avocado oil, macadamia nut oil, grapeseed oil, groundnut oil (peanut oil), sesame oil, corn oil, popcorn, whole grain wheat, cereal, oatmeal, almond oil, sunflower oil, hemp oil, and tea-oil Camellia.[107]
Polyunsaturated fat
Polyunsaturated fats are fats in which the constituent hydrocarbon chain possesses two or more carbon–carbon double bonds.[108][109] Polyunsaturated fat can be found mostly in nuts, seeds, fish, seed oils, and oysters.[108] "Unsaturated" refers to the fact that the molecules contain less than the maximum amount of hydrogen (if there were no double bonds). These materials exist as cis or trans isomers depending on the geometry of the double bond.
Saturated fats have hydrocarbon chains which can be most readily aligned. The hydrocarbon chains in trans fats align more readily than those in cis fats, but less well than those in saturated fats. In general, this means that the melting points of fats increase from cis to trans unsaturated and then to saturated. See the section about the chemical structure of fats for more information.



The position of the carbon-carbon double bonds in carboxylic acid chains in fats is designated by Greek letters.[108] The carbon atom closest to the carboxyl group is the alpha carbon, the next carbon is the beta carbon and so on. In fatty acids the carbon atom of the methyl group at the end of the hydrocarbon chain is called the omega carbon because omega is the last letter of the Greek alphabet. Omega-3 fatty acids have a double bond three carbons away from the methyl carbon, whereas omega-6 fatty acids have a double bond six carbons away from the methyl carbon. The illustration below shows the omega-6 fatty acid, linoleic acid.
While it is the nutritional aspects of polyunsaturated fats that are generally of greatest interest, these materials also have non-food applications. Drying oils, which polymerize on exposure to oxygen to form solid films, are polyunsaturated fats. The most common ones are linseed (flax seed) oil, tung oil, poppy seed oil, perilla oil, and walnut oil. These oils are used to make paints and varnishes.
Health
Potential benefits
Because of their effects in the diet, unsaturated fats (monounsaturated and polyunsaturated) are often referred to as good fats; while saturated fats are sometimes referred to as bad fats. Some fat is needed in the diet, but it is usually considered that fats should not be consumed excessively, unsaturated fats should be preferred, and saturated fats in particular should be limited.[110][111][112][113]
In preliminary research, omega-3 fatty acids in algal oil, fish oil, fish and seafood have been shown to lower the risk of heart attacks.[114] Other preliminary research indicates that omega-6 fatty acids in sunflower oil and safflower oil may also reduce the risk of cardiovascular disease.[115]
Among omega-3 fatty acids, neither long-chain nor short-chain forms were consistently associated with breast cancer risk. High levels of docosahexaenoic acid (DHA), however, the most abundant omega-3 polyunsaturated fatty acid in erythrocyte (red blood cell) membranes, were associated with a reduced risk of breast cancer.[116] The DHA obtained through the consumption of polyunsaturated fatty acids is positively associated with cognitive and behavioral performance.[117] In addition DHA is vital for the grey matter structure of the human brain, as well as retinal stimulation and neurotransmission.[108]
Contrary to conventional advice, an evaluation of evidence from 1966-1973 pertaining to the health impacts of replacing dietary saturated fat with linoleic acid found that participants in the group doing so had increased rates of death from all causes, coronary heart disease, and cardiovascular disease.[118] Although this evaluation was disputed by many scientists,[119] it fueled debate over worldwide dietary advice to substitute polyunsaturated fats for saturated fats.[120]
Pregnancy
Polyunsaturated fat supplementation does not decrease the incidence of pregnancy-related disorders, such as hypertension or preeclampsia, but may increase the length of gestation slightly and decreased the incidence of early premature births.[108]
Expert panels in the United States and Europe recommend that pregnant and lactating women consume higher amounts of polyunsaturated fats than the general population to enhance the DHA status of the fetus and newborn.[108]
Cancer
Results from observational clinical trials on polyunsaturated fat intake and cancer have been inconsistent and vary by numerous factors of cancer incidence, including gender and genetic risk.[114] Some studies have shown associations between higher intakes and/or blood levels of polyunsaturated fat omega-3s and a decreased risk of certain cancers, including breast and colorectal cancer, while other studies found no associations with cancer risk.[114][121]
Food sources
Food sources of polyunsaturated fats include:[108][122]
| Food source (100g) | Polyunsaturated fat (g) |
|---|---|
| Walnuts | 47 |
| Canola Oil | 34 |
| Sunflower seeds | 33 |
| Sesame Seeds | 26 |
| Chia Seeds | 23.7 |
| Unsalted Peanuts | 16 |
| Peanut Butter | 14.2 |
| Avocado Oil | 13.5 [123] |
| Olive Oil | 11 |
| Safflower Oil | 12.82[124] |
| Seaweed | 11 |
| Sardines | 5 |
| Soybeans | 7 |
| Tuna | 14 |
| Wild Salmon | 17.3 |
| Whole Grain Wheat | 9.7 |
Trans fat

| Types of fats in food |
|---|
| Components |
| Manufactured fats |
Trans fat, also called trans-unsaturated fatty acids or trans fatty acids, is a type of unsaturated fat that occurs in small amounts in meat and milk fat.[125] It became widely produced as an unintentional byproduct in the industrial processing of vegetable and fish oils in the early 20th century for use in margarine[126] and later also in snack food, packaged baked goods, and for frying fast food.[127][128]
Fats contain long hydrocarbon chains, which can be either unsaturated, i.e., have double bonds, or saturated, i.e., have no double bonds. In nature, unsaturated fatty acids generally have cis as opposed to trans configurations.[129] In food production, liquid cis-unsaturated fats such as vegetable oils are hydrogenated to produce saturated fats, which have more desirable physical properties: e.g., they melt at a desirable temperature (30–40 °C; 86–104 °F); and extend the shelf-life of food. Partial hydrogenation of the unsaturated fat converts some of the cis double bonds into trans double bonds by an isomerization reaction with the catalyst used for the hydrogenation, which yields a trans fat.[127][128]
Although trans fats are edible, consuming trans fats has been shown to increase the risk of coronary artery disease in part by raising levels of low-density lipoprotein (LDL, often termed "bad cholesterol"), lowering levels of high-density lipoprotein (HDL, often termed "good cholesterol"), increasing triglycerides in the bloodstream and promoting systemic inflammation.[130][131]
Trans fats also occur naturally, e.g., the vaccenic acid in breast milk, and some isomers of conjugated linoleic acid (CLA). These trans fats occur naturally in meat and dairy products from ruminants. Butter, for example, contains about 3% trans fat.[132] Two Canadian studies have shown that vaccenic acid could be beneficial compared to hydrogenated vegetable shortening, or a mixture of pork lard and soy fat, by lowering total LDL and triglyceride levels.[133][134][135][134] A study by the US Department of Agriculture showed that vaccenic acid raises both HDL and LDL cholesterol, whereas industrial trans fats only raise LDL with no beneficial effect on HDL.[136]
In light of recognized evidence and scientific agreement, nutritional authorities consider all trans fats equally harmful for health and recommend that their consumption be reduced to trace amounts.[137][138][139][140][141] The World Health Organization recommended that trans fats make up no more than 0.9% of a person's diet in 2003[142] and, in 2018, introduced a 6-step guide to eliminate industrially-produced trans-fatty acids from the global food supply.[143]
In many countries, there are legal limits to trans fat content. Trans fats levels can be reduced or eliminated by switching to saturated fats such as lard, palm oil, or fully hydrogenated fats, or by using interesterified fat. Other alternative formulations can also allow unsaturated fats to be used to replace saturated or partially hydrogenated fats. Hydrogenated oil is not a synonym for trans fat: complete hydrogenation removes all unsaturated fats.
History

Nobel laureate Paul Sabatier worked in the late 1890s to develop the chemistry of hydrogenation, which enabled the margarine, oil hydrogenation, and synthetic methanol industries.[144] Whereas Sabatier considered hydrogenation of only vapors, the German chemist Wilhelm Normann showed in 1901 that liquid oils could be hydrogenated, and patented the process in 1902.[145][146][147] During the years 1905–1910, Normann built a fat-hardening facility in the Herford company. At the same time, the invention was extended to a large-scale plant in Warrington, England, at Joseph Crosfield & Sons, Limited. It took only two years until the hardened fat could be successfully produced in the plant in Warrington, commencing production in the autumn of 1909. The initial year's production totalled nearly 3,000 tonnes.[148] In 1909, Procter & Gamble acquired the United States rights to the Normann patent;[149] in 1911, they began marketing the first hydrogenated shortening, Crisco (composed largely of partially hydrogenated cottonseed oil). Further success came from the marketing technique of giving away free cookbooks in which every recipe called for Crisco.
Normann's hydrogenation process made it possible to stabilize affordable whale oil or fish oil for human consumption, a practice kept secret to avoid consumer distaste.[148]
Before 1910, dietary fats in industrialized nations consisted mostly of butterfat, beef tallow, and lard. During Napoleon's reign in France in the early 19th century, a type of margarine was invented to feed the troops using tallow and buttermilk. It was not accepted in the United States. In the early 20th century, soybeans began to be imported into the United States as a source of protein; soybean oil was a by-product. What to do with that oil became an issue. At the same time, there was not enough butterfat available for consumers. The method of hydrogenating fat and turning a liquid fat into a solid one had been discovered, and now the ingredients (soybeans) and the need (shortage of butter) were there. Later, the means for storage, the refrigerator, was a factor in trans fat development. The fat industry found that hydrogenated fats provided some special features to margarines, which allowed margarine, unlike butter, to be taken out of a refrigerator and immediately spread on bread. By some minor changes to the chemical composition of hydrogenated fat, such hydrogenated fat was found to provide superior baking properties compared to lard. Margarine made from hydrogenated soybean oil began to replace butterfat. Hydrogenated fat such as Crisco and Spry, sold in England, began to replace butter and lard in baking bread, pies, cookies, and cakes in 1920.[150]
Production of hydrogenated fats increased steadily until the 1960s, as processed vegetable fats replaced animal fats in the United States and other Western countries. At first, the argument was a financial one due to lower costs; advocates also said that the unsaturated trans fats of margarine were healthier than the saturated fats of butter.[151]
A University of Guelph research group has found a way to mix oils (such as olive, soybean, and canola), water, monoglycerides, and fatty acids to form a "cooking fat" that acts the same way as trans and saturated fats.[152][153]
Chemistry
In chemical terms, trans fat is a fat (lipid) molecule that contains one or more double bonds in trans geometric configuration.
A double bond may exhibit one of two possible configurations: trans or cis. In trans configuration, the carbon chain extends from opposite sides of the double bond, whereas, in cis configuration, the carbon chain extends from the same side of the double bond. The trans molecule is a straighter molecule. The cis molecule is bent.[154]
| Trans (Elaidic acid) | Unsaturated (Oleic acid) | Saturated (Stearic acid) |
|---|---|---|
| Elaidic acid is the main trans unsaturated fatty acid often found in partially hydrogenated vegetable oils.[155] | Oleic acid is an unsaturated fatty acid making up 55–80% of olive oil.[156] | Stearic acid is a saturated fatty acid found in animal fats and is the intended product in full hydrogenation. Stearic acid is neither unsaturated nor trans because it has no carbon-carbon double bonds. |
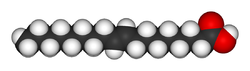 |
 |
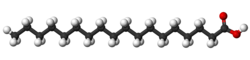
|
 |
||
| These fatty acids are geometric isomers (structurally identical except for the arrangement of the double bond). | This fatty acid contains no carbon-carbon double bonds and is not isomeric with the prior two. | |
A fatty acid is characterized as either saturated or unsaturated based on the presence of double bonds in its structure. If the molecule contains no double bonds, it is said to be saturated; otherwise, it is unsaturated to some degree.[157][158]
Only unsaturated fats can be trans or cis fat, since only a double bond can be locked to these orientations. Saturated fatty acids are never called trans fats because they have no double bonds. Thus, all their bonds are freely rotatable. Other types of fatty acids, such as crepenynic acid, which contains a triple bond, are rare and of no nutritional significance.[citation needed]
Carbon atoms are tetravalent, forming four covalent bonds with other atoms, whereas hydrogen atoms bond with only one other atom. In saturated fatty acids, each carbon atom (besides the last) is connected to its two neighbour carbon atoms and to two hydrogen atoms. In unsaturated fatty acids, the carbon atoms that are missing a hydrogen atom are joined by double bonds rather than single bonds so that each carbon atom still participates in four bonds.

Hydrogenation of an unsaturated fatty acid refers to the addition of hydrogen atoms to the acid, causing double bonds to become single ones, as carbon atoms acquire new hydrogen partners (to maintain four bonds per carbon atom). Full hydrogenation results in a molecule containing the maximum amount of hydrogen (in other words, the conversion of an unsaturated fatty acid into a saturated one). Partial hydrogenation results in the addition of hydrogen atoms at some of the empty positions, with a corresponding reduction in the number of double bonds. Typical commercial hydrogenation is partial to obtain a malleable mixture of fats that is solid at room temperature, but melts during baking, or consumption.
In most naturally occurring unsaturated fatty acids, the hydrogen atoms are on the same side of the double bonds of the carbon chain (cis configuration – from the Latin, meaning "on the same side"). However, partial hydrogenation reconfigures most of the double bonds that do not become chemically saturated, twisting them so that the hydrogen atoms end up on different sides of the chain. This type of configuration is called trans, from the Latin, meaning "across".[159] The trans configuration is the lower energy form, and is favored when catalytically equilibrated as a side reaction in hydrogenation.
The same molecule, containing the same number of atoms, with a double bond in the same location, can be either a trans or a cis fatty acid depending on the configuration of the double bond. For example, oleic acid and elaidic acid are both unsaturated fatty acids with the chemical formula C9H17C9H17O2.[160] They both have a double bond located midway along the carbon chain. It is the configuration of this bond that sets them apart. The configuration has implications for the physical-chemical properties of the molecule. The trans configuration is straighter, while the cis configuration is noticeably kinked as can be seen from the three-dimensional representation shown above.
The trans fatty acid elaidic acid has different chemical and physical properties, owing to the slightly different bond configuration. It has a much higher melting point, 45 °C, than oleic acid, 13.4 °C, due to the ability of the trans molecules to pack more tightly, forming a solid that is more difficult to break apart.[160] This notably means that it is a solid at human body temperatures.
In the sense of food production, however, the goal is not necessarily to simply change the configuration of double bonds while maintaining the same ratios of hydrogen to carbon; rather, the goal is to decrease the number of double bonds (when a fatty acid molecule contains more than one double bond it is classified as "polyunsaturated") by increasing the amount of hydrogen (and, thus, single bonds) in the fatty acid. This subsequent lesser degree of unsaturation (and, simultaneously, greater degree of saturation) thereby changes the consistency of the fatty acid by way of allowing its molecules to more greatly compress and congeal and in turn thereby makes it less prone to rancidity (in which free radicals attack double bonds). In this second sense of the goal being to simply reduce the degree of unsaturation in an unsaturated fatty acid, the production of trans fatty acids is thus an undesirable side effect of partial hydrogenation.
Catalytic partial hydrogenation necessarily produces trans-fats, because of the reaction mechanism. In the first reaction step, one hydrogen is added, with the other, coordinatively unsaturated, carbon being attached to the catalyst. The second step is the addition of hydrogen to the remaining carbon, producing a saturated fatty acid. The first step is reversible, such that the hydrogen is readsorbed on the catalyst and the double bond is re-formed. The intermediate with only one hydrogen added contains no double bond and can freely rotate. Thus, the double bond can re-form as either cis or trans, of which trans is favored, regardless the starting material. Complete hydrogenation also hydrogenates any produced trans fats to give saturated fats.
Researchers at the United States Department of Agriculture have investigated whether hydrogenation can be achieved without the side effect of trans fat production. They varied the pressure under which the chemical reaction was conducted – applying 1400 kPa (200 psi) of pressure to soybean oil in a 2-liter vessel while heating it to between 140 °C and 170 °C. The standard 140 kPa (20 psi) process of hydrogenation produces a product of about 40% trans fatty acid by weight, compared to about 17% using the high-pressure method. Blended with unhydrogenated liquid soybean oil, the high-pressure-processed oil produced margarine containing 5 to 6% trans fat. Based on current U.S. labeling requirements (see below), the manufacturer could claim the product was free of trans fat.[161] The level of trans fat may also be altered by modification of the temperature and the length of time during hydrogenation.
Trans fat levels may be measured. Measurement techniques include chromatography (by silver ion chromatography on thin layer chromatography plates, or small high-performance liquid chromatography columns of silica gel with bonded phenylsulfonic acid groups whose hydrogen atoms have been exchanged for silver ions). The role of silver lies in its ability to form complexes with unsaturated compounds. Gas chromatography and mid-infrared spectroscopy are other methods in use.
Presence in food
| Food type | Trans fat content |
|---|---|
| shortenings | 10g to 33 g |
| margarine, spreads | 0.2[163] to 26 g |
| butter | 2g to 7 g |
| whole milk | 0.07g to 0.1 g |
| breads/cake products | 0.1g to 10 g |
| cookies and crackers | 1g to 8 g |
| salty snacks | 0g to 4 g |
| cake frostings, sweets | 0.1g to 7 g |
| animal fat | 0g to 5 g[142] |
| ground beef | 1 g |
A type of trans fat occurs naturally in the milk and body fat of ruminants (such as cattle and sheep) at a level of 2–5% of total fat.[142] Natural trans fats, which include conjugated linoleic acid (CLA) and vaccenic acid, originate in the rumen of these animals. CLA has two double bonds, one in the cis configuration and one in trans, which makes it simultaneously a cis- and a trans-fatty acid.{{[164]}}
Animal-based fats were once the only trans fats consumed, but by far the largest amount of trans fat consumed today is created by the processed food industry as a side effect of partially hydrogenating unsaturated plant fats (generally vegetable oils). These partially hydrogenated fats have displaced natural solid fats and liquid oils in many areas, the most notable ones being in the fast food, snack food, fried food, and baked goods industries.[165]
Partially hydrogenated oils have been used in food for many reasons. Hydrogenation increases product shelf life and decreases refrigeration requirements. Many baked foods require semi-solid fats to suspend solids at room temperature; partially hydrogenated oils have the right consistency to replace animal fats such as butter and lard at lower cost. They are also an inexpensive alternative to other semi-solid oils such as palm oil.[citation needed]
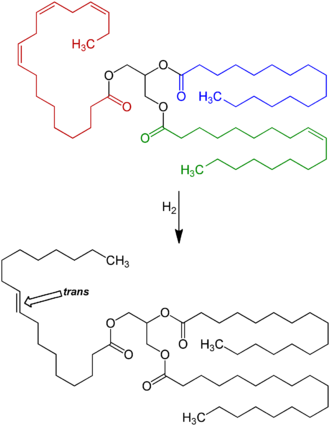
Up to 45% of the total fat in those foods containing man-made trans fats formed by partially hydrogenating plant fats may be trans fat.[142] Baking shortenings, unless reformulated, contain around 30% trans fats compared to their total fats. High-fat dairy products such as butter contain about 4%. Margarines not reformulated to reduce trans fats may contain up to 15% trans fat by weight,[166] but some reformulated ones are less than 1% trans fat.
It has been established that trans fats in human breast milk fluctuate with maternal consumption of trans fat, and that the amount of trans fats in the bloodstream of breastfed infants fluctuates with the amounts found in their milk. In 1999, reported percentages of trans fats (compared to total fats) in human milk ranged from 1% in Spain, 2% in France, 4% in Germany, and 7% in Canada and the United States.[167]
Trans fats are used in shortenings for deep-frying in restaurants, as they can be used for longer than most conventional oils before becoming rancid. In the early 21st century, non-hydrogenated vegetable oils that have lifespans exceeding that of the frying shortenings became available.[168] As fast-food chains routinely use different fats in different locations, trans fat levels in fast food can have large variations. For example, an analysis of samples of McDonald's French fries collected in 2004 and 2005 found that fries served in New York City contained twice as much trans fat as in Hungary, and 28 times as much as in Denmark, where trans fats are restricted. At KFC, the pattern was reversed, with Hungary's product containing twice the trans fat of the New York product. Even within the United States there was variation, with fries in New York containing 30% more trans fat than those from Atlanta.[169]
Nutritional guidelines
The National Academy of Sciences (NAS) advises the United States and Canadian governments on nutritional science for use in public policy and product labeling programs. Their 2002 Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids[170] contains their findings and recommendations regarding consumption of trans fat (summary).
Their recommendations are based on two key facts. First, "trans fatty acids are not essential and provide no known benefit to human health",[130] whether of animal or plant origin.[171] Second, while both saturated and trans fats increase levels of LDL, trans fats also lower levels of HDL;[172] thus increasing the risk of coronary artery disease. The NAS is concerned "that dietary trans fatty acids are more deleterious with respect to coronary artery disease than saturated fatty acids".[172] This analysis is supported by a 2006 New England Journal of Medicine (NEJM) scientific review that states "from a nutritional standpoint, the consumption of trans fatty acids results in considerable potential harm but no apparent benefit."[173]
Because of these facts and concerns, the NAS has concluded there is no safe level of trans fat consumption. There is no adequate level, recommended daily amount or tolerable upper limit for trans fats. This is because any incremental increase in trans fat intake increases the risk of coronary artery disease.[172]
Despite this concern, the NAS dietary recommendations have not included eliminating trans fat from the diet. This is because trans fat is naturally present in many animal foods in trace quantities, and thus its removal from ordinary diets might introduce undesirable side effects and nutritional imbalances if proper nutritional planning is not undertaken. The NAS has, thus, "recommended that trans fatty acid consumption be as low as possible while consuming a nutritionally adequate diet".[174] Like the NAS, the World Health Organization has tried to balance public health goals with a practical level of trans fat consumption, recommending in 2003 that trans fats be limited to less than 1% of overall energy intake.[142]
The US National Dairy Council has asserted that the trans fats present in animal foods are of a different type than those in partially hydrogenated oils, and do not appear to exhibit the same negative effects.[175] While a recent scientific review agrees with the conclusion (stating that "the sum of the current evidence suggests that the Public health implications of consuming trans fats from ruminant products are relatively limited"), it cautions that this may be due to the low consumption of trans fats from animal sources compared to artificial ones.[173]
More recent inquiry (independent of the dairy industry) has found in a 2008 Dutch meta-analysis that all trans fats, regardless of natural or artificial origin equally raise LDL and lower HDL levels.[176] Other studies though have shown different results when it comes to animal based trans fats like conjugated linoleic acid (CLA). Although CLA is known for its anticancer properties, researchers have also found that the cis-9, trans-11 form of CLA can reduce the risk for cardiovascular disease and help fight inflammation.[177][178]
Health risks
Partially hydrogenated vegetable oils have been an increasingly significant part of the human diet for about 100 years (in particular, since the later half of the 20th century and where more processed foods are consumed),[179] and some deleterious effects of trans fat consumption are scientifically accepted, forming the basis of the health guidelines discussed above.
The exact biochemical process by which trans fats produce specific health problems are a topic of continuing research. Intake of dietary trans fat perturbs the body's ability to metabolize essential fatty acids (EFAs, including Omega-3) leading to changes in the phospholipid fatty acid composition of the arterial walls, thereby raising risk of coronary artery disease.[180]
While the mechanisms through which trans fatty acids contribute to coronary artery disease are fairly well understood, the mechanism for their effects on diabetes is still under investigation. They may impair the metabolism of long-chain polyunsaturated fatty acids (LCPUFAs),[181] but maternal pregnancy trans fatty acid intake has been inversely associated with LCPUFAs levels in infants at birth thought to underlie the positive association between breastfeeding and intelligence.[182]
High intake of trans fatty acids can lead to many health problems throughout one's life.[183] They are abundant in fast food restaurants.[179] They are consumed in greater quantities by people who lack access to a diet consisting of fewer hydrogenated fats, or who often consume fast food. A diet high in trans fats can contribute to obesity, high blood pressure, and higher risk for heart disease[citation needed]. Trans fat has also been implicated in the development of Type 2 diabetes.[184]
Coronary artery disease
The primary health risk identified for trans fat consumption is an elevated risk of coronary artery disease (CAD).[185] A 1994 study estimated that over 30,000 cardiac deaths per year in the United States are attributable to the consumption of trans fats.[186] By 2006 upper estimates of 100,000 deaths were suggested.[187] A comprehensive review of studies of trans fats published in 2006 in the New England Journal of Medicine reports a strong and reliable connection between trans fat consumption and CAD, concluding that "On a per-calorie basis, trans fats appear to increase the risk of CAD more than any other macronutrient, conferring a substantially increased risk at low levels of consumption (1 to 3% of total energy intake)".[173][173]
The major evidence for the effect of trans fat on CAD comes from the Nurses' Health Study – a cohort study that has been following 120,000 female nurses since its inception in 1976. In this study, Hu and colleagues analyzed data from 900 coronary events from the study's population during 14 years of followup. He determined that a nurse's CAD risk roughly doubled (relative risk of 1.93, CI: 1.43 to 2.61) for each 2% increase in trans fat calories consumed (instead of carbohydrate calories). By contrast, for each 5% increase in saturated fat calories (instead of carbohydrate calories) there was a 17% increase in risk (relative risk of 1.17, CI: 0.97 to 1.41). "The replacement of saturated fat or trans unsaturated fat by cis (unhydrogenated) unsaturated fats was associated with larger reductions in risk than an isocaloric replacement by carbohydrates."[188] Hu also reports on the benefits of reducing trans fat consumption. Replacing 2% of food energy from trans fat with non-trans unsaturated fats more than halves the risk of CAD (53%). By comparison, replacing a larger 5% of food energy from saturated fat with non-trans unsaturated fats reduces the risk of CAD by 43%.[188]
Another study considered deaths due to CAD, with consumption of trans fats being linked to an increase in mortality, and consumption of polyunsaturated fats being linked to a decrease in mortality.[185][189]
There are two accepted tests that measure an individual's risk for coronary artery disease, both blood tests. The first considers ratios of two types of cholesterol, the other the amount of a cell-signalling cytokine called C-reactive protein. The ratio test is more accepted, while the cytokine test may be more powerful but is still being studied.[185] The effect of trans fat consumption has been documented on each as follows:
- Cholesterol ratio: This ratio compares the levels of LDL to HDL. Trans fat behaves like saturated fat by raising the level of LDL, but, unlike saturated fat, it has the additional effect of decreasing levels of HDL. The net increase in LDL/HDL ratio with trans fat is approximately double that due to saturated fat.[190][191][192] (Higher ratios are worse.) One randomized crossover study published in 2003 comparing the effect of eating a meal on blood lipids of (relatively) cis and trans fat rich meals showed that cholesteryl ester transfer (CET) was 28% higher after the trans meal than after the cis meal and that lipoprotein concentrations were enriched in apolipoprotein(a) after the trans meals.[193]
- C-reactive protein (CRP): A study of over 700 nurses showed that those in the highest quartile of trans fat consumption had blood levels of CRP that were 73% higher than those in the lowest quartile.[194]
Other health risks
There are suggestions that the negative consequences of trans fat consumption go beyond the cardiovascular risk. In general, there is much less scientific consensus asserting that eating trans fat specifically increases the risk of other chronic health problems:
- Alzheimer's Disease: A study published in Archives of Neurology in February 2003 suggested that the intake of both trans fats and saturated fats promote the development of Alzheimer disease,[195] although not confirmed in an animal model.[196] It has been found that trans fats impaired memory and learning in middle-age rats. The trans-fat eating rats' brains had fewer proteins critical to healthy neurological function. Inflammation in and around the hippocampus, the part of the brain responsible for learning and memory. These are the exact types of changes normally seen at the onset of Alzheimer's, but seen after six weeks, even though the rats were still young.[197]
- Cancer: There is no scientific consensus that consuming trans fats significantly increases cancer risks across the board.[185] The American Cancer Society states that a relationship between trans fats and cancer "has not been determined."[198] One study has found a positive connection between trans fat and prostate cancer.[199] However, a larger study found a correlation between trans fats and a significant decrease in high-grade prostate cancer.[200] An increased intake of trans fatty acids may raise the risk of breast cancer by 75%, suggest the results from the French part of the European Prospective Investigation into Cancer and Nutrition.[201][202]
- Diabetes: There is a growing concern that the risk of type 2 diabetes increases with trans fat consumption.[185] However, consensus has not been reached.[173] For example, one study found that risk is higher for those in the highest quartile of trans fat consumption.[203] Another study has found no diabetes risk once other factors such as total fat intake and BMI were accounted for.[204]
- Obesity: Research indicates that trans fat may increase weight gain and abdominal fat, despite a similar caloric intake.[205] A 6-year experiment revealed that monkeys fed a trans fat diet gained 7.2% of their body weight, as compared to 1.8% for monkeys on a mono-unsaturated fat diet.[206][207] Although obesity is frequently linked to trans fat in the popular media,[208] this is generally in the context of eating too many calories; there is not a strong scientific consensus connecting trans fat and obesity, although the 6-year experiment did find such a link, concluding that "under controlled feeding conditions, long-term TFA consumption was an independent factor in weight gain. TFAs enhanced intra-abdominal deposition of fat, even in the absence of caloric excess, and were associated with insulin resistance, with evidence that there is impaired post-insulin receptor binding signal transduction."[207]
- Liver dysfunction: Trans fats are metabolized differently by the liver than other fats and interfere with delta 6 desaturase. Delta 6 desaturase is an enzyme involved in converting essential fatty acids to arachidonic acid and prostaglandins, both of which are important to the functioning of cells.[209]
- Infertility in women: One 2007 study found, "Each 2% increase in the intake of energy from trans unsaturated fats, as opposed to that from carbohydrates, was associated with a 73% greater risk of ovulatory infertility...".[210]
- Major depressive disorder: Spanish researchers analysed the diets of 12,059 people over six years and found that those who ate the most trans fats had a 48 per cent higher risk of depression than those who did not eat trans fats.[211] One mechanism may be trans-fats' substitution for docosahexaenoic acid (DHA) levels in the orbitofrontal cortex (OFC). Very high intake of trans-fatty acids (43% of total fat) in mice from 2 to 16 months of age was associated with lowered DHA levels in the brain (p=0.001).[196] When the brains of 15 major depressive subjects who had committed suicide were examined post-mortem and compared against 27 age-matched controls, the suicidal brains were found to have 16% less (male average) to 32% less (female average) DHA in the OFC. The OFC controls reward, reward expectation, and empathy (all of which are reduced in depressive mood disorders) and regulates the limbic system.[212]
- Behavioral irritability and aggression: a 2012 observational analysis of subjects of an earlier study found a strong relation between dietary trans fat acids and self-reported behavioral aggression and irritability, suggesting but not establishing causality.[213]
- Diminished memory: In a 2015 article, researchers re-analyzing results from the 1999-2005 UCSD Statin Study argue that "greater dietary trans fatty acid consumption is linked to worse word memory in adults during years of high productivity, adults age <45".[214]
- Acne: According to a 2015 study, trans fats are one of several components of Western pattern diets which promote acne, along with carbohydrates with high glycemic load such as refined sugars or refined starches, milk and dairy products, and saturated fats, while omega-3 fatty acids, which reduce acne, are deficient in Western pattern diets.[215]
Public response and regulation
See also
Further reading
- Dijkstra, Albert; Hamilton, Richard J.; Wolf, Hamm, eds. (2008). Trans Fatty Acids. Blackwell. ISBN 978-1-4051-5691-2.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - Jang ES, Jung MY, Min DB (2005). "Hydrogenation for Low Trans and High Conjugated Fatty Acids" (PDF). Comprehensive Reviews in Food Science and Food Safety. 1. Archived from the original (PDF) on 17 December 2008.
External links
- "Ban the Trans: These Sorry Lipids Should Go Away"
- Center for Science in the Public Interest Trans Fat Page
- Harvard School of Public Health webpage on trans-fat
- "Labeling & Nutrition – Guidance for Industry: Trans Fatty Acids in Nutrition Labeling, Nutrient Content Claims, Health Claims; Small Entity Compliance Guide". Center for Food Safety and Applied Nutrition. August 2003. Archived from the original on 26 October 2013. Retrieved 6 April 2014.
- Federal Register – 68 FR 41433 11 July 2003: Food Labeling: Trans Fatty Acids in Nutrition Labeling, Nutrient Content Claims, and Health Claims
Nutritional and health aspects
The benefits and risks of dietary fats have been the object of much study, and are still highly controversial topics.[216][217][218][219]
Essential fatty acids
There are two essential fatty acids (EFAs) in human nutrition: alpha-linolenic acid (an omega-3 fatty acid) and linoleic acid (an omega-6 fatty acid).[220][8] Other lipids needed by the body can be synthesized from these and other fats.
Saturated vs. unsaturated fats
Studies have found that replacing saturated fats with cis unsaturated fats in the diet reduces risk of cardiovascular disease. For example, a 2020 systematic review of randomized control trials by the Cochrane Library concluded: "Lifestyle advice to all those at risk of cardiovascular disease and to lower risk population groups should continue to include permanent reduction of dietary saturated fat and partial replacement by unsaturated fats."[221]
Cis vs. trans fats
Numerous studies have also found that consumption of trans fats increases risk of cardiovascular disease.[220][8] The Harvard School of Public Health advises that replacing trans fats and saturated fats with cis monounsaturated and polyunsaturated fats is beneficial for health.[222]
Interesterification
Some studies have investigated the health effects of insteresterified (IE) fats, by comparing diets with IE and non-IE fats with the same overall fatty acid composition.[223]
Several experimental studies in humans found no statistical difference on fasting blood lipids between a with large amounts of IE fat, having 25-40% C16:0 or C18:0 on the 2-position, and a similar diet with non-IE fat, having only 3-9% C16:0 or C18:0 on the 2-position.[224][225][226] A negative result was obtained also in a study that compared the effects on blood cholesterol levels of an IE fat product mimicking cocoa butter and the real non-IE product.[227][228][229][230][231][232][233]
A 2007 study funded by the Malaysian Palm Oil Board[234] claimed that replacing natural palm oil by other interesterified or partial hydrogenated fats caused adverse health effects, such as higher LDL/HDL ratio and plasma glucose levels. However, these effects could be attributed to the higher percentage of saturated acids in the IE and partially hydrogenated fats, rather than to the IE process itself.[235][236]
Fat digestion and metabolism
Fats are broken down in the healthy body to release their constituents, glycerol and fatty acids. Glycerol itself can be converted to glucose by the liver and so become a source of energy. Fats and other lipids are broken down in the body by enzymes called lipases produced in the pancreas.
Many cell types can use either glucose or fatty acids as a source of energy for metabolism. In particular, heart and skeletal muscle prefer fatty acids.[citation needed] Despite long-standing assertions to the contrary, fatty acids can also be used as a source of fuel for brain cells through mitochondrial oxidation. [237]
See also
- Animal fat
- Carbohydrate
- Dieting
- Fat content of milk
- Food composition data
- Lipid
- Omega-3 fatty acid
- Omega-6 fatty acid
- Trans fat
- Vegetable oil
- Yellow grease
- List of saturated fatty acids
- List of vegetable oils
- Trans fat
- Food groups
- Food guide pyramid
- Healthy diet
- Diet and heart disease
- Fast food
- Junk food
- Advanced glycation endproduct
- ANGPTL4
- Iodine value
- Framingham Heart Study
- Seven Countries Study
- Ancel Keys
- D. Mark Hegsted
- Western pattern diet
- Iodine value – a chemical analysis method to determine the proportion of unsaturated fat.
- List of unsaturated fatty acids
Further reading
- Feinman RD (October 2010). "Saturated fat and health: recent advances in research". Lipids. 45 (10): 891–2. doi:10.1007/s11745-010-3446-8. PMC 2974200. PMID 20827513.
- Howard BV, Van Horn L, Hsia J, Manson JE, Stefanick ML, Wassertheil-Smoller S, et al. (2006). "Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial". Journal of the American Medical Association. 295 (6): 655–66. doi:10.1001/jama.295.6.655. PMID 16467234.
- Zelman K (May 2011). "The great fat debate: a closer look at the controversy-questioning the validity of age-old dietary guidance". Journal of the American Dietetic Association. 111 (5): 655–8. doi:10.1016/j.jada.2011.03.026. PMID 21515106.
External links
References
- ^ a b c d Entry for "fat" in the online Merriam-Webster disctionary, sense 3.2. Accessed on 2020-08-09
- ^ a b c Thomas A. B. Sanders (2016): "The Role of Fats in Human Diet". Pages 1-20 of Functional Dietary Lipids. Woodhead/Elsevier, 332 pages. ISBN 978-1-78242-247-1doi:10.1016/B978-1-78242-247-1.00001-6
- ^ "Macronutrients: the Importance of Carbohydrate, Protein, and Fat". McKinley Health Center. University of Illinois at Urbana–Champaign. Retrieved 20 September 2014.
- ^ "Introduction to Energy Storage". Khan Academy.
- ^ Anna Ohtera, Yusaku Miyamae, Naomi Nakai, Atsushi Kawachi, Kiyokazu Kawada, Junkyu Han, Hiroko Isoda, Mohamed Neffati, Toru Akita, Kazuhiro Maejima, Seiji Masuda, Taiho Kambe, Naoki Mori, Kazuhiro Irie, and Masaya Nagao (2013): "Identification of 6-octadecynoic acid from a methanol extract of Marrubium vulgare L. as a peroxisome proliferator-activated receptor γ agonist". Biochemical and Biophysical Research Communications, volume 440, issue 2, pages 204-209. doi:10.1016/j.bbrc.2013.09.003
- ^ N. Koeniger and H. J. Veith (1983): "Glyceryl-1,2-dioleate-3-palmitate, a brood pheromone of the honey bee (Apis mellifera L.)". Experientia, volume 39, pages 1051–1052 doi:10.1007/BF01989801
- ^ Institute of Shortenings and Edible oils (2006). "Food Fats and oils" (PDF). Archived from the original (PDF) on 2007-03-26. Retrieved 2009-02-19.
- ^ a b c Government of the United Kingdom (1996): "Schedule 7: Nutrition labelling". In Food Labelling Regulations 1996'. Accessed on 2020-08-09.
- ^ "The human proteome in adipose - The Human Protein Atlas". www.proteinatlas.org. Retrieved 2017-09-12.
- ^ a b Reece, Jane; Campbell, Neil (2002). Biology. San Francisco: Benjamin Cummings. pp. 69–70. ISBN 978-0-8053-6624-2.
- ^ "Saturated fats". American Heart Association. 2014. Retrieved 1 March 2014.
- ^ "Top food sources of saturated fat in the US". Harvard University School of Public Health. 2014. Retrieved 1 March 2014.
- ^ "Saturated, Unsaturated, and Trans Fats". choosemyplate.gov. 2020.
- ^ "Saturated Fat". American Heart Association. 2020.
- ^ "What are "oils"?". ChooseMyPlate.gov, US Department of Agriculture. 2015. Archived from the original on 9 June 2015. Retrieved 13 June 2015.
- ^ Hooper L, Martin N, Abdelhamid A, Davey Smith G (June 2015). "Reduction in saturated fat intake for cardiovascular disease". The Cochrane Database of Systematic Reviews. 6 (6): CD011737. doi:10.1002/14651858.CD011737. PMID 26068959.
- ^ a b c d e f Sacks FM, Lichtenstein AH, Wu JH, Appel LJ, Creager MA, Kris-Etherton PM, Miller M, Rimm EB, Rudel LL, Robinson JG, Stone NJ, Van Horn LV (July 2017). "Dietary Fats and Cardiovascular Disease: A Presidential Advisory From the American Heart Association". Circulation. 136 (3): e1–e23. doi:10.1161/CIR.0000000000000510. PMID 28620111. S2CID 367602.
- ^ de Souza RJ, Mente A, Maroleanu A, Cozma AI, Ha V, Kishibe T, Uleryk E, Budylowski P, Schünemann H, Beyene J, Anand SS (August 2015). "Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: systematic review and meta-analysis of observational studies". BMJ. 351 (Aug 11): h3978. doi:10.1136/bmj.h3978. PMC 4532752. PMID 26268692.
- ^ Ramsden CE, Zamora D, Leelarthaepin B, Majchrzak-Hong SF, Faurot KR, Suchindran CM, et al. (February 2013). "Use of dietary linoleic acid for secondary prevention of coronary heart disease and death: evaluation of recovered data from the Sydney Diet Heart Study and updated meta-analysis". BMJ. 346: e8707. doi:10.1136/bmj.e8707. PMC 4688426. PMID 23386268.
- ^ Ramsden CE, Zamora D, Majchrzak-Hong S, Faurot KR, Broste SK, Frantz RP, Davis JM, Ringel A, Suchindran CM, Hibbeln JR (April 2016). "Re-evaluation of the traditional diet-heart hypothesis: analysis of recovered data from Minnesota Coronary Experiment (1968-73)". BMJ. 353: i1246. doi:10.1136/bmj.i1246. PMC 4836695. PMID 27071971.
- ^ "USDA National Nutrient Database for Standard Reference, Release 20". United States Department of Agriculture. 2007. Archived from the original on 2016-04-14.
- ^ a b Hooper L, Martin N, Jimoh OF, Kirk C, Foster E, Abdelhamid AS (2020). "Reduction in saturated fat intake for cardiovascular disease". Cochrane Database of Systematic Reviews (Systematic review). 5: CD011737. doi:10.1002/14651858.CD011737.pub2. ISSN 1465-1858. PMC 7388853. PMID 32428300.
- ^ Graham I, Atar D, Borch-Johnsen K, Boysen G, Burell G, Cifkova R, et al. (2007). "European guidelines on cardiovascular disease prevention in clinical practice: executive summary". European Heart Journal. 28 (19): 2375–2414. doi:10.1093/eurheartj/ehm316. PMID 17726041.
- ^ Labarthe, Darwin (2011). "Chapter 17 What Causes Cardiovascular Diseases?". Epidemiology and prevention of cardiovascular disease: a global challenge (2nd ed.). Jones and Bartlett Publishers. ISBN 978-0-7637-4689-6.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Kris-Etherton PM, Innis S (September 2007). "Position of the American Dietetic Association and Dietitians of Canada: Dietary Fatty Acids". Journal of the American Dietetic Association. 107 (9): 1599–1611 [1603]. doi:10.1016/j.jada.2007.07.024. PMID 17936958.
- ^ "Food Fact Sheet - Cholesterol" (PDF). British Dietetic Association. Retrieved 3 May 2012.
- ^ a b "Cardiovascular Disease Risk Factors". World Heart Federation. 30 May 2017. Retrieved 2012-05-03.
- ^ "Lower your cholesterol". National Health Service. Retrieved 2012-05-03.
- ^ "Nutrition Facts at a Glance - Nutrients: Saturated Fat". Food and Drug Administration. 2009-12-22. Retrieved 2012-05-03.
- ^ "Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol". European Food Safety Authority. 2010-03-25. Retrieved 3 May 2012.
- ^ "Healthy diet Fact sheet N°394". May 2015. Retrieved 12 August 2015.
- ^ Faculty of Public Health of the Royal Colleges of Physicians of the United Kingdom. Position Statement on Fat [Retrieved 2011-01-25].
- ^ Report of a Joint WHO/FAO Expert Consultation (2003). "Diet, Nutrition and the Prevention of Chronic Diseases" (PDF). World Health Organization. Retrieved 2011-03-11.
- ^ "Cholesterol". Irish Heart Foundation. Retrieved 2011-02-28.
- ^ U.S. Department of Agriculture and U.S. Department of Health and Human Services (December 2010). Dietary Guidelines for Americans, 2010 (PDF) (7th ed.). Washington, DC: U.S. Government Printing Office.
- ^ Cannon, Christopher; O'Gara, Patrick (2007). Critical Pathways in Cardiovascular Medicine (2nd ed.). Lippincott Williams & Wilkins. p. 243.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Clarke R, Frost C, Collins R, Appleby P, Peto R (1997). "Dietary lipids and blood cholesterol: quantitative meta-analysis of metabolic ward studies". BMJ (Clinical Research Ed.). 314 (7074): 112–7. doi:10.1136/bmj.314.7074.112. PMC 2125600. PMID 9006469.
- ^ Bucher HC, Griffith LE, Guyatt GH (February 1999). "Systematic review on the risk and benefit of different cholesterol-lowering interventions". Arteriosclerosis, Thrombosis, and Vascular Biology. 19 (2): 187–195. doi:10.1161/01.atv.19.2.187. PMID 9974397.
- ^ a b Lewington S, Whitlock G, Clarke R, Sherliker P, Emberson J, Halsey J, Qizilbash N, Peto R, Collins R (December 2007). "Blood cholesterol and vascular mortality by age, sex, and blood pressure: a meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths". Lancet. 370 (9602): 1829–39. doi:10.1016/S0140-6736(07)61778-4. PMID 18061058. S2CID 54293528.
- ^ Labarthe, Darwin (2011). "Chapter 11 Adverse Blood Lipid Profile". Epidemiology and prevention of cardiovascular disease: a global challenge (2 ed.). Jones and Bartlett Publishers. p. 290. ISBN 978-0-7637-4689-6.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Labarthe, Darwin (2011). "Chapter 11 Adverse Blood Lipid Profile". Epidemiology and prevention of cardiovascular disease: a global challenge (2nd ed.). Jones and Bartlett Publishers. p. 277. ISBN 978-0-7637-4689-6.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Thijssen MA, Mensink RP (2005). "Fatty acids and atherosclerotic risk". Atherosclerosis: Diet and Drugs. Handbook of Experimental Pharmacology. Vol. 170. Springer. pp. 165–94. doi:10.1007/3-540-27661-0_5. ISBN 978-3-540-22569-0. PMID 16596799.
- ^ Boyd NF, Stone J, Vogt KN, Connelly BS, Martin LJ, Minkin S (November 2003). "Dietary fat and breast cancer risk revisited: a meta-analysis of the published literature". British Journal of Cancer. 89 (9): 1672–1685. doi:10.1038/sj.bjc.6601314. PMC 2394401. PMID 14583769.
- ^ a b Hanf V, Gonder U (2005-12-01). "Nutrition and primary prevention of breast cancer: foods, nutrients and breast cancer risk". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 123 (2): 139–149. doi:10.1016/j.ejogrb.2005.05.011. PMID 16316809.
- ^ Lof M, Weiderpass E (February 2009). "Impact of diet on breast cancer risk". Current Opinion in Obstetrics and Gynecology. 21 (1): 80–85. doi:10.1097/GCO.0b013e32831d7f22. PMID 19125007. S2CID 9513690.
- ^ Freedman LS, Kipnis V, Schatzkin A, Potischman N (Mar–Apr 2008). "Methods of Epidemiology: Evaluating the Fat–Breast Cancer Hypothesis – Comparing Dietary Instruments and Other Developments". Cancer Journal (Sudbury, Mass.). 14 (2): 69–74. doi:10.1097/PPO.0b013e31816a5e02. PMC 2496993. PMID 18391610.
- ^ Lin OS (2009). "Acquired risk factors for colorectal cancer". Cancer Epidemiology. Methods in Molecular Biology. Vol. 472. pp. 361–72. doi:10.1007/978-1-60327-492-0_16. ISBN 978-1-60327-491-3. PMID 19107442.
- ^ Huncharek M, Kupelnick B (2001). "Dietary fat intake and risk of epithelial ovarian cancer: a meta-analysis of 6,689 subjects from 8 observational studies". Nutrition and Cancer. 40 (2): 87–91. doi:10.1207/S15327914NC402_2. PMID 11962260. S2CID 24890525.
- ^ a b Männistö S, Pietinen P, Virtanen MJ, Salminen I, Albanes D, Giovannucci E, Virtamo J (December 2003). "Fatty acids and risk of prostate cancer in a nested case-control study in male smokers". Cancer Epidemiology, Biomarkers & Prevention. 12 (12): 1422–8. PMID 14693732.
- ^ a b c Crowe FL, Allen NE, Appleby PN, Overvad K, Aardestrup IV, Johnsen NF, Tjønneland A, Linseisen J, Kaaks R, Boeing H, Kröger J, Trichopoulou A, Zavitsanou A, Trichopoulos D, Sacerdote C, Palli D, Tumino R, Agnoli C, Kiemeney LA, Bueno-de-Mesquita HB, Chirlaque MD, Ardanaz E, Larrañaga N, Quirós JR, Sánchez MJ, González CA, Stattin P, Hallmans G, Bingham S, Khaw KT, Rinaldi S, Slimani N, Jenab M, Riboli E, Key TJ (November 2008). "Fatty acid composition of plasma phospholipids and risk of prostate cancer in a case-control analysis nested within the European Prospective Investigation into Cancer and Nutrition". The American Journal of Clinical Nutrition. 88 (5): 1353–63. doi:10.3945/ajcn.2008.26369 (inactive 2020-08-23). PMID 18996872.
{{cite journal}}: CS1 maint: DOI inactive as of August 2020 (link) - ^ a b Kurahashi N, Inoue M, Iwasaki M, Sasazuki S, Tsugane AS (April 2008). "Dairy product, saturated fatty acid, and calcium intake and prostate cancer in a prospective cohort of Japanese men". Cancer Epidemiology, Biomarkers & Prevention. 17 (4): 930–7. doi:10.1158/1055-9965.EPI-07-2681. PMID 18398033. S2CID 551427.
- ^ Corwin RL, Hartman TJ, Maczuga SA, Graubard BI (2006). "Dietary saturated fat intake is inversely associated with bone density in humans: Analysis of NHANES III". The Journal of Nutrition. 136 (1): 159–165. doi:10.1093/jn/136.1.159. PMID 16365076. S2CID 4443420.
- ^ see the article Food pyramid (nutrition) for more information.
- ^ "Choosing foods with healthy fats". Health Canada. 2018-10-10. Retrieved 2019-09-24.
- ^ "Cut Down on Saturated Fats" (PDF). United States Department of Health and Human Services. Retrieved 2019-09-24.
- ^ "Fat: the facts". United Kingdom's National Health Service. 2018-04-27. Retrieved 2019-09-24.
- ^ "Fat". Australia's National Health and Medical Research Council and Department of Health and Ageing. 2012-09-24. Retrieved 2019-09-24.
- ^ "Getting the Fats Right!". Singapore's Ministry of Health. Retrieved 2019-09-24.
- ^ "Health Diet". India's Ministry of Health and Family Welfare. Retrieved 2019-09-24.
- ^ "Eating and Activity Guidelines for New Zealand Adults" (PDF). New Zealand's Ministry of Health. Retrieved 2019-09-24.
- ^ "Know More about Fat". Hong Kong's Department of Health. Retrieved 2019-09-24.
- ^ a b Joint WHO/FAO Expert Consultation (2003). Diet, Nutrition and the Prevention of Chronic Diseases (WHO technical report series 916) (PDF). World Health Organization. pp. 81–94. ISBN 978-92-4-120916-8. Retrieved 2016-04-04.
- ^ "Trends in Intake of Energy, Protein, Carbohydrate, Fat, and Saturated Fat — United States, 1971–2000". Centers for Disease Control. 2004. Archived from the original on 2008-12-01.
- ^ Lichtenstein AH, Appel LJ, Brands M, Carnethon M, Daniels S, Franch HA, Franklin B, Kris-Etherton P, Harris WS, Howard B, Karanja N, Lefevre M, Rudel L, Sacks F, Van Horn L, Winston M, Wylie-Rosett J (July 2006). "Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee". Circulation. 114 (1): 82–96. doi:10.1161/CIRCULATIONAHA.106.176158. PMID 16785338. S2CID 647269.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Smith SC, Jackson R, Pearson TA, Fuster V, Yusuf S, Faergeman O, Wood DA, Alderman M, Horgan J, Home P, Hunn M, Grundy SM (June 2004). "Principles for national and regional guidelines on cardiovascular disease prevention: a scientific statement from the World Heart and Stroke Forum" (PDF). Circulation. 109 (25): 3112–21. doi:10.1161/01.CIR.0000133427.35111.67. PMID 15226228.
- ^ "Dietary Guidelines for Americans" (PDF). United States Department of Agriculture. 2005.
- ^ Eat less saturated fat
- ^ Fats explained
- ^ German JB, Dillard CJ (September 2004). "Saturated fats: what dietary intake?". American Journal of Clinical Nutrition. 80 (3): 550–559. doi:10.1093/ajcn/80.3.550. PMID 15321792.
- ^ Zelman K (2011). "The Great Fat Debate: A Closer Look at the Controversy—Questioning the Validity of Age-Old Dietary Guidance". Journal of the American Dietetic Association. 111 (5): 655–658. doi:10.1016/j.jada.2011.03.026. PMID 21515106.
- ^ Catapano AL, Reiner Z, De Backer G, Graham I, Taskinen MR, Wiklund O, et al. (July 2011). "ESC/EAS Guidelines for the management of dyslipidaemias: the Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS)". Atherosclerosis. 217 Suppl 1 (14): S1-44. doi:10.1016/j.atherosclerosis.2011.06.012. hdl:10138/307445. PMID 21723445.
- ^ Mozaffarian D, Rimm EB, Herrington DM (November 2004). "Dietary fats, carbohydrate, and progression of coronary atherosclerosis in postmenopausal women". The American Journal of Clinical Nutrition. 80 (5): 1175–84. doi:10.1093/ajcn/80.5.1175. PMC 1270002. PMID 15531663.
- ^ Leibovitz B, Hu ML, Tappel AL (January 1990). "Dietary supplements of vitamin E, beta-carotene, coenzyme Q10 and selenium protect tissues against lipid peroxidation in rat tissue slices". The Journal of Nutrition. 120 (1): 97–104. doi:10.1093/jn/120.1.97. PMID 2303916.
- ^ Fats and sugars. BBC Health, retrieved 2013-04-07
- ^ a b Storlien LH, Baur LA, Kriketos AD, Pan DA, Cooney GJ, Jenkins AB, et al. (June 1996). "Dietary fats and insulin action". Diabetologia. 39 (6): 621–31. doi:10.1007/BF00418533. PMID 8781757.
- ^ Aizpurua-Olaizola O, Ormazabal M, Vallejo A, Olivares M, Navarro P, Etxebarria N, et al. (January 2015). "Optimization of supercritical fluid consecutive extractions of fatty acids and polyphenols from Vitis vinifera grape wastes". Journal of Food Science. 80 (1): E101-7. doi:10.1111/1750-3841.12715. PMID 25471637.
- ^ Breuer B, Stuhlfauth T, Fock HP (July 1987). "Separation of fatty acids or methyl esters including positional and geometric isomers by alumina argentation thin-layer chromatography". Journal of Chromatographic Science. 25 (7): 302–6. doi:10.1093/chromsci/25.7.302. PMID 3611285.
- ^ "Thrive Culinary Algae Oil". Retrieved 7 January 2019.
- ^ a b c d e f Anderson D. "Fatty acid composition of fats and oils" (PDF). Colorado Springs: University of Colorado, Department of Chemistry. Retrieved April 8, 2017.
- ^ "NDL/FNIC Food Composition Database Home Page". United States Department of Agriculture, Agricultural Research Service. Retrieved May 21, 2013.
- ^ "Basic Report: 04042, Oil, peanut, salad or cooking". USDA. Archived from the original on March 9, 2016. Retrieved 16 January 2015.
- ^ "Oil, vegetable safflower, oleic". nutritiondata.com. Condé Nast. Retrieved 10 April 2017.
- ^ "Oil, vegetable safflower, linoleic". nutritiondata.com. Condé Nast. Retrieved 10 April 2017.
- ^ "Oil, vegetable, sunflower". nutritiondata.com. Condé Nast. Retrieved 27 September 2010.
- ^ USDA Basic Report Cream, fluid, heavy whipping
- ^ "Nutrition And Health". The Goose Fat Information Service.
- ^ "Egg, yolk, raw, fresh". nutritiondata.com. Condé Nast. Retrieved 24 August 2009.
- ^ "09038, Avocados, raw, California". National Nutrient Database for Standard Reference, Release 26. United States Department of Agriculture, Agricultural Research Service. Archived from the original on January 10, 2014. Retrieved 14 August 2014.
- ^ a b Hulbert AJ, Else PL (August 1999). "Membranes as possible pacemakers of metabolism". Journal of Theoretical Biology. 199 (3): 257–74. doi:10.1006/jtbi.1999.0955. PMID 10433891.
- ^ Hulbert AJ, Faulks S, Buttemer WA, Else PL (November 2002). "Acyl composition of muscle membranes varies with body size in birds". The Journal of Experimental Biology. 205 (Pt 22): 3561–9. PMID 12364409.
- ^ Hulbert AJ (July 2003). "Life, death and membrane bilayers". The Journal of Experimental Biology. 206 (Pt 14): 2303–11. doi:10.1242/jeb.00399. PMID 12796449.
- ^ Raynard RS, Cossins AR (May 1991). "Homeoviscous adaptation and thermal compensation of sodium pump of trout erythrocytes". The American Journal of Physiology. 260 (5 Pt 2): R916–24. doi:10.1152/ajpregu.1991.260.5.R916. PMID 2035703.
- ^ Vessby B, Uusitupa M, Hermansen K, Riccardi G, Rivellese AA, Tapsell LC, Nälsén C, Berglund L, Louheranta A, Rasmussen BM, Calvert GD, Maffetone A, Pedersen E, Gustafsson IB, Storlien LH (March 2001). "Substituting dietary saturated for monounsaturated fat impairs insulin sensitivity in healthy men and women: The KANWU Study". Diabetologia. 44 (3): 312–9. doi:10.1007/s001250051620. PMID 11317662.
- ^ Lovejoy JC (October 2002). "The influence of dietary fat on insulin resistance". Current Diabetes Reports. 2 (5): 435–40. doi:10.1007/s11892-002-0098-y. PMID 12643169.
- ^ Fukuchi S, Hamaguchi K, Seike M, Himeno K, Sakata T, Yoshimatsu H (June 2004). "Role of fatty acid composition in the development of metabolic disorders in sucrose-induced obese rats". Experimental Biology and Medicine. 229 (6): 486–93. doi:10.1177/153537020422900606. PMID 15169967.
- ^ Kien CL, Bunn JY, Tompkins CL, Dumas JA, Crain KI, Ebenstein DB, Koves TR, Muoio DM (April 2013). "Substituting dietary monounsaturated fat for saturated fat is associated with increased daily physical activity and resting energy expenditure and with changes in mood". The American Journal of Clinical Nutrition. 97 (4): 689–97. doi:10.3945/ajcn.112.051730. PMC 3607650. PMID 23446891.
- ^ "You Can Control Your Cholesterol: A Guide to Low-Cholesterol Living". MerckSource. Archived from the original on 2009-03-03. Retrieved 2018-01-02.
- ^ "Monounsaturated Fat". American Heart Association. Archived from the original on 2018-03-07. Retrieved 2018-04-19.
- ^ Pala V, Krogh V, Muti P, Chajès V, Riboli E, Micheli A, Saadatian M, Sieri S, Berrino F (July 2001). "Erythrocyte membrane fatty acids and subsequent breast cancer: a prospective Italian study". Journal of the National Cancer Institute. 93 (14): 1088–95. doi:10.1093/jnci/93.14.1088. PMID 11459870.
- ^ Sanchez-Bayle M, Gonzalez-Requejo A, Pelaez MJ, Morales MT, Asensio-Anton J, Anton-Pacheco E (February 2008). "A cross-sectional study of dietary habits and lipid profiles. The Rivas-Vaciamadrid study". European Journal of Pediatrics. 167 (2): 149–54. doi:10.1007/s00431-007-0439-6. PMID 17333272.
- ^ Dinu M, Pagliai G, Casini A, Sofi F (January 2018). "Mediterranean diet and multiple health outcomes: an umbrella review of meta-analyses of observational studies and randomised trials". European Journal of Clinical Nutrition. 72 (1): 30–43. doi:10.1038/ejcn.2017.58. PMID 28488692.
- ^ Martinez-Lacoba R, Pardo-Garcia I, Amo-Saus E, Escribano-Sotos F (October 2018). "Mediterranean diet and health outcomes: a systematic meta-review". European Journal of Public Health. 28 (5): 955–961. doi:10.1093/eurpub/cky113. PMID 29992229.
- ^ https://www.thrivealgae.com/our-product/. Retrieved 7 January 2019.
{{cite web}}: Missing or empty|title=(help) - ^ Abdullah MM, Jew S, Jones PJ (February 2017). "Health benefits and evaluation of healthcare cost savings if oils rich in monounsaturated fatty acids were substituted for conventional dietary oils in the United States". Nutrition Reviews. 75 (3): 163–174. doi:10.1093/nutrit/nuw062. PMC 5914363. PMID 28158733.
- ^ Huth PJ, Fulgoni VL, Larson BT (November 2015). "A systematic review of high-oleic vegetable oil substitutions for other fats and oils on cardiovascular disease risk factors: implications for novel high-oleic soybean oils". Advances in Nutrition. 6 (6): 674–93. doi:10.3945/an.115.008979. PMC 4642420. PMID 26567193.
- ^ Board on Agriculture and Renewable Resources Commission on Natural Resources and Food and Nutrition Board, Assembly of Life Sciences, National Research Council (1976). Fat content and composition of animal products: proceedings of a symposium, Washington, D.C., December 12-13, 1974. Washington: National Academy of Sciences. ISBN 978-0-309-02440-2. PMID 25032409.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Aizpurua-Olaizola O, Ormazabal M, Vallejo A, Olivares M, Navarro P, Etxebarria N, Usobiaga A (January 2015). "Optimization of supercritical fluid consecutive extractions of fatty acids and polyphenols from Vitis vinifera grape wastes". Journal of Food Science. 80 (1): E101-7. doi:10.1111/1750-3841.12715. PMID 25471637.
- ^ a b c d e f g "Essential Fatty Acids". Micronutrient Information Center, Oregon State University, Corvallis, OR. May 2014. Retrieved 24 May 2017.
- ^ "Omega-3 fatty acids, fish oil, alpha-linolenic acid". Mayo Clinic. 2017. Retrieved 24 May 2017.
- ^ "Fats explained" (PDF). HEART UK – The Cholesterol Charity. Retrieved 20 February 2019.
- ^ "Key Recommendations: Components of Healthy Eating Patterns". Dietary Guidelines 2015-2020. Retrieved 20 February 2019.
- ^ "Live Well, Eat well, Fat: the facts". NHS. Retrieved 20 February 2019.
- ^ "Dietary Guidelines for Indians - A Manual" (PDF). Indian Council of Medical Research, National Institute of Nutrition. Archived from the original (PDF) on 2018-12-22. Retrieved 2019-02-20.
- ^ a b c "Omega-3 Fatty Acids and Health: Fact Sheet for Health Professionals". US National Institutes of Health, Office of Dietary Supplements. 2 November 2016. Retrieved 5 April 2017.
- ^ Willett WC (September 2007). "The role of dietary n-6 fatty acids in the prevention of cardiovascular disease". Journal of Cardiovascular Medicine. 8 Suppl 1: S42-5. doi:10.2459/01.JCM.0000289275.72556.13. PMID 17876199.
- ^ Pala V, Krogh V, Muti P, Chajès V, Riboli E, Micheli A, Saadatian M, Sieri S, Berrino F (July 2001). "Erythrocyte membrane fatty acids and subsequent breast cancer: a prospective Italian study". Journal of the National Cancer Institute. 93 (14): 1088–95. doi:10.1093/jnci/93.14.1088. PMID 11459870.
- ^ van de Rest O, Geleijnse JM, Kok FJ, van Staveren WA, Dullemeijer C, Olderikkert MG, Beekman AT, de Groot CP (August 2008). "Effect of fish oil on cognitive performance in older subjects: a randomized, controlled trial". Neurology. 71 (6): 430–8. doi:10.1212/01.wnl.0000324268.45138.86. PMID 18678826.
- ^ Ramsden CE, Zamora D, Leelarthaepin B, Majchrzak-Hong SF, Faurot KR, Suchindran CM, Ringel A, Davis JM, Hibbeln JR (February 2013). "Use of dietary linoleic acid for secondary prevention of coronary heart disease and death: evaluation of recovered data from the Sydney Diet Heart Study and updated meta-analysis". BMJ. 346: e8707. doi:10.1136/bmj.e8707. PMC 4688426. PMID 23386268.
- ^ Interview: Walter Willett (2017). "Research Review: Old data on dietary fats in context with current recommendations: Comments on Ramsden et al. in the British Medical Journal". TH Chan School of Public Health, Harvard University, Boston. Retrieved 24 May 2017.
- ^ Weylandt KH, Serini S, Chen YQ, Su HM, Lim K, Cittadini A, Calviello G (2015). "Omega-3 Polyunsaturated Fatty Acids: The Way Forward in Times of Mixed Evidence". BioMed Research International. 2015: 143109. doi:10.1155/2015/143109. PMC 4537707. PMID 26301240.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Patterson RE, Flatt SW, Newman VA, Natarajan L, Rock CL, Thomson CA, Caan BJ, Parker BA, Pierce JP (February 2011). "Marine fatty acid intake is associated with breast cancer prognosis". The Journal of Nutrition. 141 (2): 201–6. doi:10.3945/jn.110.128777. PMC 3021439. PMID 21178081.
- ^ "National nutrient database for standard reference, release 23". United States Department of Agriculture, Agricultural Research Service. 2011. Archived from the original on 2015-03-03. Retrieved 2009-02-22.
- ^ "Vegetable oil, avocado Nutrition Facts & Calories". nutritiondata.self.com.
- ^ "United States Department of Agriculture – National Nutrient Database". 8 September 2015.
- ^ Kuhnt K, Baehr M, Rohrer C, Jahreis G (October 2011). "Trans fatty acid isomers and the trans-9/trans-11 index in fat containing foods". European Journal of Lipid Science and Technology. 113 (10): 1281–1292. doi:10.1002/ejlt.201100037. PMC 3229980. PMID 22164125.
- ^ Gormley JJ, Juturu V (2010). "Partially Hydrogenated Fats in the US Diet and Their Role in Disease". In De Meester F, Zibadi S, Watson RR (eds.). Modern Dietary Fat Intakes in Disease Promotion. Nutrition and Health. Totowa, NJ: Humana Press. pp. 85–94. doi:10.1007/978-1-60327-571-2_5. ISBN 9781603275712.
{{cite encyclopedia}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ a b "About Trans Fat and Partially Hydrogenated Oils" (PDF). Center for Science in the Public Interest.
- ^ a b "Tentative Determination Regarding Partially Hydrogenated Oils". Federal Register. 8 November 2013. 2013-26854, Vol. 78, No. 217. Archived from the original on 6 April 2014. Retrieved 8 November 2013.
- ^ Martin CA, Milinsk MC, Visentainer JV, Matsushita M, de-Souza NE (June 2007). "Trans fatty acid-forming processes in foods: a review". Anais da Academia Brasileira de Ciencias. 79 (2): 343–50. doi:10.1590/S0001-37652007000200015. PMID 17625687.
- ^ a b Food and nutrition board, institute of medicine of the national academies (2005). Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). National Academies Press. pp. 423. doi:10.17226/10490. ISBN 978-0-309-08525-0.
- ^ "Trans fat: Avoid this cholesterol double whammy". Mayo Foundation for Medical Education and Research (MFMER). Retrieved 10 December 2007.
- ^ "National Nutrient Database for Standard Reference Release 28". United States Department of Agriculture.[permanent dead link]
- ^ Trans Fats From Ruminant Animals May Be Beneficial – Health News. redOrbit (8 September 2011). Retrieved 22 January 2013.
- ^ a b Bassett CM, Edel AL, Patenaude AF, McCullough RS, Blackwood DP, Chouinard PY, et al. (January 2010). "Dietary vaccenic acid has antiatherogenic effects in LDLr-/- mice". The Journal of Nutrition. 140 (1): 18–24. doi:10.3945/jn.109.105163. PMID 19923390.
- ^ Wang Y, Jacome-Sosa MM, Vine DF, Proctor SD (20 May 2010). "Beneficial effects of vaccenic acid on postprandial lipid metabolism and dyslipidemia: Impact of natural trans-fats to improve CVD risk". Lipid Technology. 22 (5): 103–106. doi:10.1002/lite.201000016.
- ^ David J. Baer, PhD. US Department of Agriculture, Agricultural Research Service, Beltsville Human Nutrition Research Laboratory. New Findings on Dairy Trans Fat and Heart Disease Risk, IDF World Dairy Summit 2010, 8–11 November 2010. Auckland, New Zealand
- ^ EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA) (2010). "Scientific opinion on dietary reference values for fats". EFSA Journal. 8 (3): 1461. doi:10.2903/j.efsa.2010.1461.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ UK Scientific Advisory Committee on Nutrition (2007). "Update on trans fatty acids and health, Position Statement" (PDF). Archived from the original (PDF) on 10 December 2010.
- ^ Brouwer IA, Wanders AJ, Katan MB (March 2010). "Effect of animal and industrial trans fatty acids on HDL and LDL cholesterol levels in humans--a quantitative review". PLOS ONE. 5 (3): e9434. Bibcode:2010PLoSO...5.9434B. doi:10.1371/journal.pone.0009434. PMC 2830458. PMID 20209147.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Trans fat". It's your health. Health Canada. Dec 2007. Archived from the original on 20 April 2012.
- ^ "EFSA sets European dietary reference values for nutrient intakes" (Press release). European Food Safety Authority. 26 March 2010.
- ^ a b c d e Trans Fat Task Force (June 2006). TRANSforming the Food Supply. ISBN 0-662-43689-X. Retrieved 7 January 2007.
- ^ "WHO plan to eliminate industrially-produced trans-fatty acids from global food supply" (Press release). World Health Organization. 14 May 2018.
- ^ Nobel Lectures, Chemistry, 1901–1921. Elsevier. 1966. Reprinted online: "Paul Sabatier, The Nobel Prize in Chemistry 1912". Nobel Foundation. Retrieved 7 January 2007.
- ^ de 141029 Process for converting unsaturated fatty acids or their glycerides into saturated compounds
- ^ gb 190301515 Process for converting unsaturated fatty acids or their glycerides into saturated compounds
- ^ Patterson HB (1998). "Hydrogenation" (PDF). Sci Lecture Papers Series. Archived from the original (PDF) on 26 September 2007. Retrieved 7 January 2007.
- ^ a b "Wilhelm Normann und die Geschichte der Fetthärtung von Martin Fiedler, 2001". 20 December 2011. Archived from the original on 1 October 2011. Retrieved 14 August 2007.
- ^ Shurtleff, William; Aoyagi, Akiko. "History of Soybeans and Soyfoods: 1100 B.C. to the 1980s". Archived from the original on 18 October 2005.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Kummerow, Fred A. (2008). Cholesterol Won't Kill You – But Trans Fat Could. Trafford Publishing. ISBN 978-1-4251-3808-0.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Ascherio A, Stampfer MJ, Willett WC. "Trans fatty acids and coronary heart disease". Archived from the original on 3 September 2006. Retrieved 14 September 2006.
- ^ Hadzipetros, Peter (25 January 2007). "Trans Fats Headed for the Exit". CBC News.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Spencelayh, Michael (9 January 2007). "Trans fat free future". Royal Society of Chemistry.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Udo Erasmus; Fats that heal, Fats that Kill, Alive books, 1993 edition, Pages 13-19.
- ^ Alonso L, Fontecha J, Lozada L, Fraga MJ, Juárez M (May 1999). "Fatty acid composition of caprine milk: major, branched-chain, and trans fatty acids". Journal of Dairy Science. 82 (5): 878–84. doi:10.3168/jds.S0022-0302(99)75306-3. hdl:10261/113439. PMID 10342226.
- ^ Thomas, Alfred (2002). "Fats and Fatty Oils". Ullmann's Encyclopedia of Industrial Chemistry. Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_173. ISBN 978-3-527-30673-2.
{{cite encyclopedia}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Casimir C. Akoh; David B. Min., eds. (2002). Food lipids: chemistry, nutrition, and biotechnology. New York: M. Dekker. pp. 1–2. ISBN 978-0-8247-0749-1.
- ^ ""fatty acid"". IUPAC Gold book. International Union of Pure and Applied Chemistry.
- ^ Hill, John W.; Kolb, Doris K (2007). Chemistry for changing times. Pearson / Prentice Hall. ISBN 978-0136054498.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ a b "Section 7: Biochemistry". Handbook of chemistry and physics. 2007–2008 (88th ed.). Taylor and Francis. 2007. Retrieved 19 November 2007.
{{cite book}}: External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help) - ^ Eller FJ, List GR, Teel JA, Steidley KR, Adlof RO (July 2005). "Preparation of spread oils meeting U.S. Food and Drug Administration Labeling requirements for trans fatty acids via pressure-controlled hydrogenation". Journal of Agricultural and Food Chemistry. 53 (15): 5982–4. doi:10.1021/jf047849+. PMID 16028984.
- ^ Tarrago-Trani MT, Phillips KM, Lemar LE, Holden JM (June 2006). "New and existing oils and fats used in products with reduced trans-fatty acid content". Journal of the American Dietetic Association. 106 (6): 867–80. doi:10.1016/j.jada.2006.03.010. PMID 16720128.
- ^ "Heart Foundation: Butter has 20 times the trans fats of marg | Australian Food News". www.ausfoodnews.com.au.
- ^ trans-fatty acid
- ^ Ashok, Chauhan; Ajit, Varma (2009). "Chapter 4: Fatty acids". A Textbook of Molecular Biotechnology. p. 181. ISBN 978-93-80026-37-4.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Hunter JE (2005). "Dietary levels of trans fatty acids" basis for health concerns and industry efforts to limit use". Nutrition Research. 25 (5): 499–513. doi:10.1016/j.nutres.2005.04.002.
- ^ Innis SM, King DJ (September 1999). "trans Fatty acids in human milk are inversely associated with concentrations of essential all-cis n-6 and n-3 fatty acids and determine trans, but not n-6 and n-3, fatty acids in plasma lipids of breast-fed infants". The American Journal of Clinical Nutrition. 70 (3): 383–90. doi:10.1093/ajcn/70.3.383. PMID 10479201.
- ^ NYC Board of Health. "Board of Health Approves Regulation to Phase Out Artificial Trans Fat: FAQ". Archived from the original on 6 October 2006. Retrieved 7 January 2007.
- ^ "What's in that french fry? Fat varies by city". NBC News. 12 April 2006. Retrieved 7 January 2007. AP story concerning Stender, S; Dyerberg, J; Astrup, A (April 2006). "High levels of industrially produced trans fat in popular fast foods". N. Engl. J. Med. 354: 1650–2. doi:10.1056/NEJMc052959. PMID 16611965.
- ^ Food and nutrition board, institute of medicine of the national academies (2005). Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academies Press. pp. i. Archived from the original on 18 September 2006.
- ^ Food and nutrition board, institute of medicine of the national academies (2005). Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academies Press. p. 447.[permanent dead link]
- ^ a b c Food and nutrition board, institute of medicine of the national academies (2005). Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). National Academies Press. p. 504.[permanent dead link]
- ^ a b c d e Mozaffarian D, Katan MB, Ascherio A, Stampfer MJ, Willett WC (April 2006). "Trans fatty acids and cardiovascular disease". The New England Journal of Medicine. 354 (15): 1601–13. doi:10.1056/NEJMra054035. PMID 16611951.
- ^ Food and nutrition board, institute of medicine of the national academies (2005). Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). National Academies Press. p. 424.[permanent dead link]
- ^ National Dairy Council (18 June 2004). "comments on 'Docket No. 2003N-0076 Food Labeling: Trans Fatty Acids in Nutrition Labeling'" (PDF). Retrieved 7 January 2007.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Brouwer IA, Wanders AJ, Katan MB (March 2010). Reitsma PH (ed.). "Effect of animal and industrial trans fatty acids on HDL and LDL cholesterol levels in humans--a quantitative review". PLOS ONE. 5 (3): e9434. Bibcode:2010PLoSO...5.9434B. doi:10.1371/journal.pone.0009434. PMC 2830458. PMID 20209147.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Tricon S, Burdge GC, Kew S, Banerjee T, Russell JJ, Jones EL, et al. (September 2004). "Opposing effects of cis-9,trans-11 and trans-10,cis-12 conjugated linoleic acid on blood lipids in healthy humans". The American Journal of Clinical Nutrition. 80 (3): 614–20. doi:10.1093/ajcn/80.3.614. PMID 15321800.
- ^ Zulet MA, Marti A, Parra MD, Martínez JA (September 2005). "Inflammation and conjugated linoleic acid: mechanisms of action and implications for human health". Journal of Physiology and Biochemistry. 61 (3): 483–94. doi:10.1007/BF03168454. PMID 16440602.
- ^ a b Valenzuela A, Morgado N (1999). "Trans fatty acid isomers in human health and in the food industry". Biological Research. 32 (4): 273–87. doi:10.4067/s0716-97601999000400007. PMID 10983247.
- ^ Kummerow FA, Zhou Q, Mahfouz MM, Smiricky MR, Grieshop CM, Schaeffer DJ (April 2004). "Trans fatty acids in hydrogenated fat inhibited the synthesis of the polyunsaturated fatty acids in the phospholipid of arterial cells". Life Sciences. 74 (22): 2707–23. doi:10.1016/j.lfs.2003.10.013. PMID 15043986.
- ^ Mojska H (2003). "Influence of trans fatty acids on infant and fetus development". Acta Microbiologica Polonica. 52 Suppl: 67–74. PMID 15058815.
- ^ Koletzko B, Decsi T (October 1997). "Metabolic aspects of trans fatty acids". Clinical Nutrition. 16 (5): 229–37. doi:10.1016/s0261-5614(97)80034-9. PMID 16844601.
- ^ Menaa F, Menaa A, Menaa B, Tréton J (June 2013). "Trans-fatty acids, dangerous bonds for health? A background review paper of their use, consumption, health implications and regulation in France". European Journal of Nutrition. 52 (4): 1289–302. doi:10.1007/s00394-012-0484-4. PMID 23269652.
- ^ Riserus, Ulf (2006). "Trans fatty acids, insulin sensitivity and type 2 diabetes". Scandinavian Journal of Food and Nutrition. 50 (4): 161–165. doi:10.1080/17482970601133114.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ a b c d e Trans Fat Task Force (June 2006). "TRANSforming the Food Supply (Appendix 9iii)". Archived from the original on 25 February 2007. Retrieved 9 January 2007.
{{cite journal}}: Cite journal requires|journal=(help) (Consultation on the health implications of alternatives to trans fatty acids: Summary of Responses from Experts) - ^ Cite error: The named reference
pmid8179036was invoked but never defined (see the help page). - ^ Zaloga GP, Harvey KA, Stillwell W, Siddiqui R (October 2006). "Trans fatty acids and coronary heart disease". Nutrition in Clinical Practice. 21 (5): 505–12. doi:10.1177/0115426506021005505. PMID 16998148.
- ^ a b Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, et al. (November 1997). "Dietary fat intake and the risk of coronary heart disease in women". The New England Journal of Medicine. 337 (21): 1491–9. doi:10.1056/NEJM199711203372102. PMID 9366580.
- ^ Oh K, Hu FB, Manson JE, Stampfer MJ, Willett WC (April 2005). "Dietary fat intake and risk of coronary heart disease in women: 20 years of follow-up of the nurses' health study". American Journal of Epidemiology. 161 (7): 672–9. doi:10.1093/aje/kwi085. PMID 15781956.
- ^ Ascherio A, Katan MB, Zock PL, Stampfer MJ, Willett WC (June 1999). "Trans fatty acids and coronary heart disease". The New England Journal of Medicine. 340 (25): 1994–8. doi:10.1056/NEJM199906243402511. PMID 10379026.
- ^ Mensink RP, Katan MB (August 1990). "Effect of dietary trans fatty acids on high-density and low-density lipoprotein cholesterol levels in healthy subjects". The New England Journal of Medicine. 323 (7): 439–45. doi:10.1056/NEJM199008163230703. PMID 2374566.
- ^ Mensink RP, Zock PL, Kester AD, Katan MB (May 2003). "Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials". The American Journal of Clinical Nutrition. 77 (5): 1146–55. doi:10.1093/ajcn/77.5.1146. PMID 12716665.
- ^ Gatto LM, Sullivan DR, Samman S (May 2003). "Postprandial effects of dietary trans fatty acids on apolipoprotein(a) and cholesteryl ester transfer". The American Journal of Clinical Nutrition. 77 (5): 1119–24. doi:10.1093/ajcn/77.5.1119. PMID 12716661.
- ^ Lopez-Garcia E, Schulze MB, Meigs JB, Manson JE, Rifai N, Stampfer MJ, et al. (March 2005). "Consumption of trans fatty acids is related to plasma biomarkers of inflammation and endothelial dysfunction". The Journal of Nutrition. 135 (3): 562–6. doi:10.1093/jn/135.3.562. PMID 15735094.
- ^ Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, et al. (February 2003). "Dietary fats and the risk of incident Alzheimer disease". Archives of Neurology. 60 (2): 194–200. doi:10.1001/archneur.60.2.194. PMID 12580703.
- ^ a b Phivilay A, Julien C, Tremblay C, Berthiaume L, Julien P, Giguère Y, Calon F (March 2009). "High dietary consumption of trans fatty acids decreases brain docosahexaenoic acid but does not alter amyloid-beta and tau pathologies in the 3xTg-AD model of Alzheimer's disease". Neuroscience. 159 (1): 296–307. doi:10.1016/j.neuroscience.2008.12.006. PMID 19135506.
- ^ Granholm AC, Bimonte-Nelson HA, Moore AB, Nelson ME, Freeman LR, Sambamurti K (June 2008). "Effects of a saturated fat and high cholesterol diet on memory and hippocampal morphology in the middle-aged rat". Journal of Alzheimer's Disease. 14 (2): 133–45. doi:10.3233/JAD-2008-14202. PMC 2670571. PMID 18560126.
- ^ American Cancer Society. "Common questions about diet and cancer". Retrieved 9 January 2007.
- ^ Chavarro J, Stampfer M, Campos H, Kurth T, Willett W, Ma J (1 April 2006). "A prospective study of blood trans fatty acid levels and risk of prostate cancer". Proc. Amer. Assoc. Cancer Res. 47 (1): 943. Retrieved 9 January 2007.
- ^ Brasky TM, Till C, White E, Neuhouser ML, Song X, Goodman P, et al. (June 2011). "Serum phospholipid fatty acids and prostate cancer risk: results from the prostate cancer prevention trial". American Journal of Epidemiology. 173 (12): 1429–39. doi:10.1093/aje/kwr027. PMC 3145396. PMID 21518693.
- ^ "Breast cancer: a role for trans fatty acids?". World Health Organization (Press release). 11 April 2008. Archived from the original on 13 April 2008.
- ^ Chajès V, Thiébaut AC, Rotival M, Gauthier E, Maillard V, Boutron-Ruault MC, et al. (June 2008). "Association between serum trans-monounsaturated fatty acids and breast cancer risk in the E3N-EPIC Study". American Journal of Epidemiology. 167 (11): 1312–20. doi:10.1093/aje/kwn069. PMC 2679982. PMID 18390841.
- ^ Hu FB, van Dam RM, Liu S (July 2001). "Diet and risk of Type II diabetes: the role of types of fat and carbohydrate". Diabetologia. 44 (7): 805–17. doi:10.1007/s001250100547. PMID 11508264.
- ^ van Dam RM, Willett WC, Rimm EB, Stampfer MJ, Hu FB (March 2002). "Dietary fat and meat intake in relation to risk of type 2 diabetes in men". Diabetes Care. 25 (3): 417–24. doi:10.2337/diacare.25.3.417. PMID 11874924.
- ^ Gosline, Anna (12 June 2006). "Why fast foods are bad, even in moderation". New Scientist. Retrieved 9 January 2007.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ "Six years of fast-food fats supersizes monkeys". New Scientist (2556): 21. 17 June 2006.
- ^ a b Kavanagh K, Jones KL, Sawyer J, Kelley K, Carr JJ, Wagner JD, Rudel LL (July 2007). "Trans fat diet induces abdominal obesity and changes in insulin sensitivity in monkeys". Obesity. 15 (7): 1675–84. doi:10.1038/oby.2007.200. PMID 17636085.
- ^ Thompson, Tommy G. "Trans Fat Press Conference". Archived from the original on 9 July 2006.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help), US Secretary of health and human services - ^ Mahfouz M (1981). "Effect of dietary trans fatty acids on the delta 5, delta 6 and delta 9 desaturases of rat liver microsomes in vivo". Acta Biologica et Medica Germanica. 40 (12): 1699–1705. PMID 7345825.
- ^ Chavarro JE, Rich-Edwards JW, Rosner BA, Willett WC (January 2007). "Dietary fatty acid intakes and the risk of ovulatory infertility". The American Journal of Clinical Nutrition. 85 (1): 231–7. doi:10.1093/ajcn/85.1.231. PMID 17209201.
- ^ Roan, Shari (28 January 2011). "Trans fats and saturated fats could contribute to depression". The Sydney Morning Herald. Retrieved 8 February 2011.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ McNamara RK, Hahn CG, Jandacek R, Rider T, Tso P, Stanford KE, Richtand NM (July 2007). "Selective deficits in the omega-3 fatty acid docosahexaenoic acid in the postmortem orbitofrontal cortex of patients with major depressive disorder". Biological Psychiatry. 62 (1): 17–24. doi:10.1016/j.biopsych.2006.08.026. PMID 17188654.
- ^ Golomb BA, Evans MA, White HL, Dimsdale JE (2012). "Trans fat consumption and aggression". PLOS ONE. 7 (3): e32175. Bibcode:2012PLoSO...732175G. doi:10.1371/journal.pone.0032175. PMC 3293881. PMID 22403632.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Golomb BA, Bui AK (2015). "A Fat to Forget: Trans Fat Consumption and Memory". PLOS ONE. 10 (6): e0128129. Bibcode:2015PLoSO..1028129G. doi:10.1371/journal.pone.0128129. PMC 4470692. PMID 26083739.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Melnik BC (15 July 2015). Weinberg J (ed.). "Linking diet to acne metabolomics, inflammation, and comedogenesis: an update". Clinical, Cosmetic and Investigational Dermatology. 8: 371–88. doi:10.2147/CCID.S69135. PMC 4507494. PMID 26203267.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Rebecca J. Donatelle (2005): Health, the Basics, 6th edition. Pearson Education, San Francisco; ISBN 978-0-13-120687-8
- ^ Frank B. Hu, JoAnn E. Manson, and Walter C. Willett (2001): "Types of dietary fat and risk of coronary heart disease: A critical review". Journal of the American College of Nutrition, volume 20, issue 1, pages 5-19. doi:10.1080/07315724.2001.10719008
- ^ Lee Hooper, Carolyn D. Summerbell, Julian P. T. Higgins, Rachel L. Thompson, Nigel E. Capps, George Davey Smith, Rudolph A. Riemersma, and Shah Ebrahim (2001): "Dietary fat intake and prevention of cardiovascular disease: systematic review". The BMJ, volume 322, pages 757-. doi:10.1136/bmj.322.7289.757
- ^ George A. Bray, Sahasporn Paeratakul, Barry M. Popkin (2004): "Dietary fat and obesity: a review of animal, clinical and epidemiological studies". Physiology & Behavior, volume 83, issue 4, pages 549-555. doi:10.1016/j.physbeh.2004.08.039
- ^ a b Dariush Mozaffarian, Martijn B. Katan, Alberto Ascherio, Meir J. Stampfer, and Walter C. Willett (2006): "Trans fatty acids and cardiovascular disease". New England Journal of Medicine, volume 354, issue 15, pages 1601–1613. doi:10.1056/NEJMra054035 PMID 16611951
- ^ Lee Hooper, Nicole Martin, Oluseyi F. Jimoh, Christian Kirk, Eve Foster, and Asmaa S. Adbelhamid (2020): "Reduction in saturated fat intake for cardiovascular disease". The Cochrane Database of Systematic Reviews, volume 5, article CD011737. PMID 32428300 doi:10.1002/14651858.CD011737.pub2
- ^ "Fats and Cholesterol", Harvard School of Public Health. Retrieved 02-11-16.
- ^ Mensink, Ronald P.; Sanders, Thomas A.; Baer, David J.; Hayes, K. C.; Howles, Philip N.; Marangoni, Alejandro (2016-07-01). "The Increasing Use of Interesterified Lipids in the Food Supply and Their Effects on Health Parameters". Advances in Nutrition. 7 (4): 719–729. doi:10.3945/an.115.009662. ISSN 2161-8313. PMC 4942855. PMID 27422506.
- ^ Zock PJ, de Vries JH, de Fouw NJ, Katan MB (1995), "Positional distribution of fatty acids in dietary triglycerides: effects on fasting blood lipoprotein concentrations in humans." (PDF), Am J Clin Nutr, vol. 61, no. 1, pp. 48–551, doi:10.1093/ajcn/61.1.48, hdl:1871/11621, PMID 7825538
- ^ Nestel PJ, Noakes M, Belling GB, et al. (1995), "Effect on plasma lipids of interesterifying a mix of edible oils." (PDF), Am J Clin Nutr, vol. 62, no. 5, pp. 950–55, doi:10.1093/ajcn/62.5.950, PMID 7572740
- ^ Meijer GW, Weststrate JA (1997), "Interesterification of fats in margarine: effect on blood lipids, blood enzymes and hemostasis parameters.", Eur J Clin Nutr, vol. 51, no. 8, pp. 527–34, doi:10.1038/sj.ejcn.1600437, PMID 11248878
- ^ Grande F, Anderson JT, Keys A (1970), "Comparison of effects of palmitic and stearic acids in the diet on serum cholesterol in man." (PDF), Am J Clin Nutr, vol. 23, no. 9, pp. 1184–93, doi:10.1093/ajcn/23.9.1184, PMID 5450836
- ^ Berry SE, Miller GJ, Sanders TA (2007), "The solid fat content of stearic acid-rich fats determines their postprandial effects." (PDF), Am J Clin Nutr, vol. 85, no. 6, pp. 1486–94, doi:10.1093/ajcn/85.6.1486, PMID 17556683
- ^ Zampelas A, Williams CM, Morgan LM, et al. (1994), "The effect of triacylglycerol fatty acids positional distribution on postprandial plasma metabolite and hormone responses in normal adult men.", Br J Nutr, vol. 71, no. 3, pp. 401–10, doi:10.1079/bjn19940147, PMID 8172869
- ^ Yli-Jokipii K, Kallio H, Schwab U, et al. (2001), "Effects of palm oil and transesterified palm oil on chylomicron and VLDL triacylglycerol structures and postprandial lipid response." (PDF), J Lipid Res, vol. 42, no. 10, pp. 1618–25, PMID 11590218
- ^ Berry SE, Woodward R, Yeoh C, Miller GJ, Sanders TA (2007), "Effect of interesterification of palmitic-acid rich tryacylglycerol on postprandial lipid and factor VII response", Lipids, 42 (4): 315–323, doi:10.1007/s11745-007-3024-x, PMID 17406926
- ^ Summers LK, Fielding BA, Herd SL, et al. (1999), "Use of structured triacylglycerols containing predominantly stearic and oleic acids to probe early events in metabolic processing of dietary fat" (PDF), J Lipid Res, vol. 40, no. 10, pp. 1890–98, PMID 10508209
- ^ Christophe AB, De Greyt WF, Delanghe JR, Huyghebaert AD (2000), "Substituting enzymically interesterified butter for native butter has no effect on lipemia or lipoproteinemia in man", Annals of Nutrition and Metabolism, 44 (2): 61–67, doi:10.1159/000012822, PMID 10970994
- ^ Sundram K, Karupaiah T, Hayes K (2007). "Stearic acid-rich interesterified fat and trans-rich fat raise the LDL/HDL ratio and plasma glucose relative to palm olein in humans" (PDF). Nutr Metab. 4: 3. doi:10.1186/1743-7075-4-3. PMC 1783656. PMID 17224066. Retrieved 2007-01-19.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Destaillats F, Moulin J, Bezelgues JB (2007), "Letter to the editor: healthy alternatives to trans fats", Nutr Metab, vol. 4, p. 10, doi:10.1186/1743-7075-4-10, PMC 1867814, PMID 17462099
{{citation}}: CS1 maint: unflagged free DOI (link) - ^ Mensink RP, Zock PL, Kester AD, Katan MB (2003), "Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials." (PDF), Am J Clin Nutr, vol. 77, no. 5, pp. 1146–1155, doi:10.1093/ajcn/77.5.1146, PMID 12716665
- ^ Panov, Alexander; Orynbayeva, Zulfiya; Vavilin, Valentin; Lyakhovich, Vyacheslav. Baranova, Ancha (ed.). "Fatty Acids in Energy Metabolism of the Central Nervous System". BioMed Research International. 2014 (The Roads to Mitochondrial Dysfunction). Hindawa.
Further reading
- Hayes, K.C. (May 2005). Dietary fat and blood lipids. Retrieved March 10, 2011.