 | |
| Clinical data | |
|---|---|
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 75-95% |
| Elimination half-life | 2-6 hours (dose: 0.5-1 g) 4-12 hours (dose: >2 g) |
| Excretion | renal (77-88%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.313 |
| Chemical and physical data | |
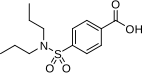
| Formula | C13H19NO4S |
| Molar mass | 285.36 g/mol g·mol−1 |
Probenecid is a uricosuric drug, primarily used in treating gout or hyperuricemia, that increases uric acid removal in the urine. One of its trade names is 'Benuryl.'
Probenecid also decreases the renal excretion of some drugs.
It was developed as an alternative to caronamide.[1]
Uses
In one study, probenecid was shown to more than double blood concentrations of oseltamivir (trade name Tamiflu), an antiviral drug used to combat influenza.[2] This is significant because nations are currently stockpiling oseltamivir in anticipation of an influenza pandemic, and there could be supply shortages.[citation needed] During World War II, probenecid was used to extend limited supplies of penicillin,[3] and is still currently used to increase antibiotic concentrations in serious infections.
It has also found use as a masking agent.[4]
Mechanism
In the kidneys it is filtered at the glomerulus, secreted in the proximal tubule and reabsorbed in the distal tubule.
Probenecid's exact mechanism is explained as follows. Gout is caused by elevated levels of uric acid in the plasma which can lead to the formation of uric acid crystals in joints, causing pain. The kidney's organic anion transporter (OAT) reclaims uric acid from the urine and returns it to the plasma. Probenecid interferes with these systems.[5] If probenecid (an organic acid) is administered to a patient, the OAT binds to probenecid instead of to uric acid, preventing the reabsorption of uric acid. As a result, more uric acid leaves the body in the urine, lowering the uric acid concentration in the plasma. This is an example of the way in which competition between substrates transported across cell membranes has been put to use in medicine.
See also
References
- ^ MASON RM (1954). "Studies on the effect of probenecid (benemid) in gout". Ann. Rheum. Dis. 13 (2): 120–30. PMC 1030399. PMID 13171805.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Hill G, Cihlar T, Oo C; et al. (2002). "The anti-influenza drug oseltamivir exhibits low potential to induce pharmacokinetic drug interactions via renal secretion-correlation of in vivo and in vitro studies". Drug Metab. Dispos. 30 (1): 13–9. PMID 11744606.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Butler D (2005). "Wartime tactic doubles power of scarce bird-flu drug". Nature. 438 (7064): 6. doi:10.1038/438006a. PMID 16267514.
- ^ Morra V, Davit P, Capra P, Vincenti M, Di Stilo A, Botrè F (2006). "Fast gas chromatographic/mass spectrometric determination of diuretics and masking agents in human urine: Development and validation of a productive screening protocol for antidoping analysis". J Chromatogr A. 1135 (2): 219–29. doi:10.1016/j.chroma.2006.09.034. PMID 17027009.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Hsyu PH, Gisclon LG, Hui AC, Giacomini KM (1988). "Interactions of organic anions with the organic cation transporter in renal BBMV". Am. J. Physiol. 254 (1 Pt 2): F56–61. PMID 2962517.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)
